Meeting Summary
At the 55th European Renal Association–European Dialysis and Transplant Association (ERA–EDTA) Congress in Copenhagen, Denmark, physicians from the USA, UK, and France presented an educational symposium entitled ‘Complex Patients May Be Better Treated with Frequent Hemodialysis: A Review and Comparison of Published Evidence and Recent European Experience’. During this symposium, leading physicians discussed the concepts underlying the prescription of frequent haemodialysis (>3 sessions per week), the role of frequent haemodialysis in managing haemodynamic instability, treating patients who require larger volume clearance due to pregnancy or obesity, and utilising frequent home haemodialysis in the palliative care setting. This report briefly summarises the symposium.
Prescribing Frequent Haemodialysis
Professor Allan Collins
Haemodialysis, as it is widely prescribed today, has created a new and unique chronic disease state in patients with end-stage renal disease who previously would not have survived. This disease state is characterised by chronic fluid overload,1,2 intense ultrafiltration during dialysis,3 and persistent hyperphosphataemia,4,5 all of which impact the heart and vessels and eventually induce cardiovascular disease.1-5
To address chronic disease, a dialysis prescription must extend beyond clearance of small solutes (e.g., urea) to target the health of organ systems. This shift in focus to monitoring a patient’s tolerance of therapy and slowing the decline of cardiovascular health represents a new paradigm in dialysis delivery; this is particularly important when dealing with complex patients, including those with uncontrolled hypertension, left ventricular hypertrophy, heart failure, hyperphosphataemia (despite prescription of phosphate binders), recurrent intradialytic hypotension, and excessive post-treatment fatigue, as well as pregnant or obese patients, and frail patients in need of supportive care.
The haemodialysis prescription can be conceptualised as a two-step approach that focusses primarily on managing fluid, thereby addressing blood pressure and cardiac geometry, and secondarily on managing solute removal.6 This model begins with setting the weekly fluid removal requirement and then the ultrafiltration rate (UFR), by way of both treatment frequency and session duration, to a rate that minimises cardiac stunning, improves stability during treatment, and minimises post-treatment fatigue. Though a conventional UFR threshold is 10 mL/hour/kg, studies have suggested that limiting UFR as much as possible may reduce all-cause and cardiovascular mortality.3,6-9 With frequent diurnal therapy for 2–3 hours per session, UFR can be lowered to 6 mL/hour/kg, and with nocturnal therapy, UFR can be lowered to 2–3 mL/hour/kg. Only after managing fluid should the haemodialysis prescription address solute removal with standardised Kt/V as a guiding metric.
Prescriptions that prioritise fluid removal can be derived in the following manner. First, an upper limit of UFR (e.g., 10 mL/hour/kg) is selected and multiplied by body weight. Second, weekly fluid intake (minus cumulative urine volume) is divided by the weight-scaled UFR limit to identify the minimum number of ultrafiltration hours per week. Third, the minimum number of ultrafiltration hours per week is divided by the number of treatments (e.g., 4, 5, or 6) that are desired during the week to target symptom-free haemodialysis and control blood pressure, ideally with minimal use of antihypertensive medication. With a UFR limit of 10 mL/hour/kg in a 70 kg male patient, for example, the maximum rate of fluid removal is 700 mL/hour. With a weekly fluid intake of 10.5 L, ≥15 hours per week of ultrafiltration are required; these could be divided among five sessions of 3 hours each or six sessions of 2 hours each. Since the uremic milieu is different during the first 90 minutes of treatment, a conventional schedule comprising three sessions of 5 hours each does not provide the same amount of clearance, despite equal hours of ultrafiltration.
After addressing fluid volume, the haemodialysis prescription must also maintain adequate solute clearance, a task largely determined by dialysate saturation. High dialysate saturation maximises the removal of small, middle, and large molecular weight solutes, even in a system that utilises low-volume dialysate (e.g., NxStage® System One™, NxStage Medical, Lawrence, Massachusetts, USA). Moreover, a system that utilises low-volume dialysate that is highly saturated during treatment will improve clearance of phosphorus, a molecule that is otherwise difficult to remove. Conventional systems with a flow rate of 500 mL/min, for example, achieve 55% urea saturation and 35% phosphorus saturation (in vivo).10 In contrast, low-volume dialysate systems with a flow rate of 200 mL/min achieve >90% urea saturation and roughly 70% phosphorus saturation (Figure 1). On nocturnal treatment with a flow rate of merely 100 mL/min, nearly 99% urea saturation and 95% phosphorus saturation can be achieved.10 Dialysate saturation is coupled with the amount of weekly total body water clearance, which varies on different modalities (Figure 2), to provide adequate clearance of solutes, including middle molecules between urea and phosphorus. Nocturnal haemodialysis maximises solute removal and minimises UFR, likely leading to improved cardiovascular outcomes.
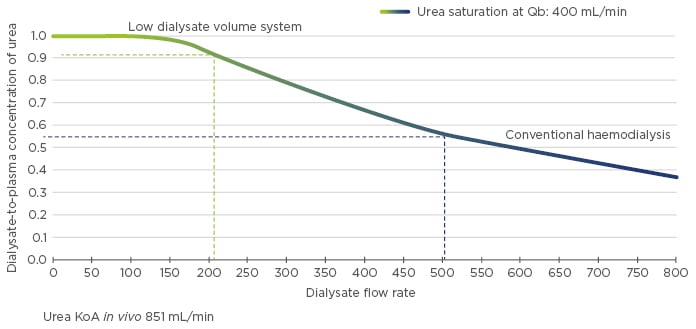
Figure 1: Dialysate-to-plasma concentration of urea as a function of dialysate flow rate.
KoA: dialyser mass transfer-area coefficient; Qb: blood flow rate.
Adapted from Leypoldt K et al.10
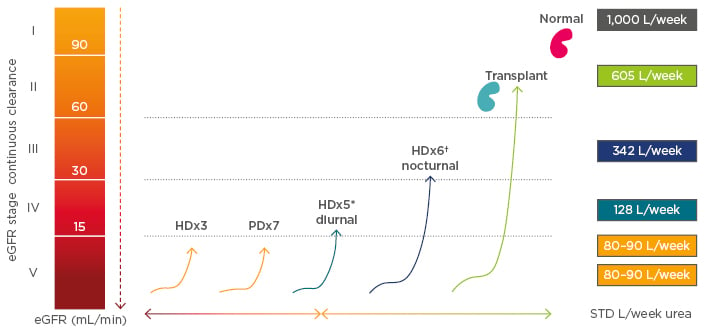
Figure 2: Volumes cleared per week among renal replacement therapies.
*30 L/session of dialysate. †60 L/session of dialysate.
eGFR: estimated glomerular filtration rate; HD: home dialysis; PD: peritoneal dialysis; STD: standard.
Adapted from Leypoldt K et al.10
A haemodialysis prescription reflecting the paradigm shift to maintaining cardiovascular health will focus first on fluid volume management, thereby minimising UFR and lowering blood pressure. Solute removal is considered next; the adequacy of removal is determined by dialysate saturation. These aims may be best achieved by a frequent treatment regimen with low-volume dialysate, which limits UFR and maximises solute clearance, thus maintaining dialysis patient health.
Managing Haemodynamic Instability with Frequent Haemodialysis
Doctor Natalie Borman
Cardiovascular disease remains the leading cause of death in dialysis patients.1,11 Through multiple physiological pathways, fluid overload is a key contributing factor to the development of heart failure on dialysis (Figure 3).1,2 Firstly, within the constraints of thrice-weekly in-centre haemodialysis, a higher UFR is needed to remove excess fluid in an overloaded patient; higher UFR can lead to cardiac and other organ system stunning during dialysis, as well as to intradialytic hypotension.12 Notably, between 5% and 15% of patients on conventional haemodialysis experience symptomatic hypotension, which is characterised by cramps, dizziness, and nausea.13,14 Organ system stunning and symptomatic hypotension both contribute to longer post-treatment recovery time, reducing quality of life and potentially leading patients to sign-off early or skip sessions entirely.15 Incomplete sessions lead to more fluid overload, thus creating a cycle of poor patient outcomes and adverse events. Secondly, chronic fluid overload contributes to uncontrolled hypertension, left ventricular hypertrophy, and heart failure, ultimately leading to increased risk of hospitalisation and death.16
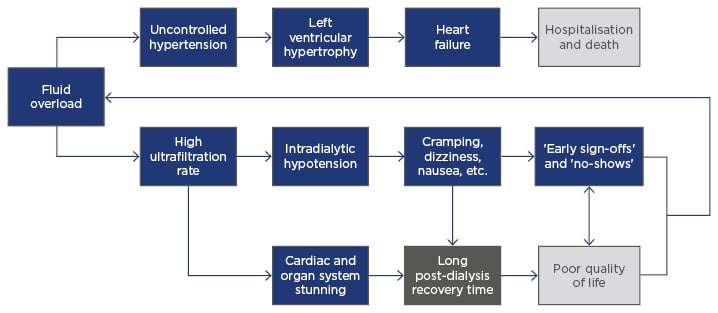
Figure 3: Causal diagram of the clinical pathophysiology of chronic fluid overload.
The thrice-weekly haemodialysis prescription became the dominant regimen because of feasibility, logistics, and cost, rather than the achievement of optimal health outcomes. The 2-day gap that is inherent in this schedule is strongly associated with increased risks of death, hospitalisation, and cardiovascular events.17-19 High UFR, which has been linked to an increased risk of all-cause and cardiovascular mortality, is prevalent in patients on conventional haemodialysis.3,7-9 Moreover, the prevalence of persistent hypertension on dialysis has not changed during the past 10 years, despite considerable improvements in small solute removal and extensive application of antihypertensive medications.2
More frequent therapy has the potential to ameliorate these symptoms and improve patients’ outcomes. Data from the Frequent Hemodialysis Network (FHN) Daily Trial show that patients prescribed more frequent haemodialysis for 1.50–2.75 hours per session have favourable outcomes for left ventricular mass, systolic blood pressure, physical health, and even death.21 Eliminating the 2-day gap not only lowers the associated risks of adverse events but also substantially reduces the likelihood of patients accumulating excess fluid between consecutive dialysis sessions.21 Furthermore, tailoring the frequency of therapy also allows for increased dialysis duration, permitting the therapy to meet weekly ultrafiltration requirements with low UFR, even in the case of fluid overload.
The KIHDNEy22 study collected data from 182 home haemodialysis patients using the NxStage System One at nine centres throughout Europe. This retrospective study followed patients for 12 months after initiating frequent home haemodialysis. Patients in the KIHDNEy cohort had a mean UFR of 6.8 mL/hour/kg at 6 and 12 months follow-up after initiating frequent therapy, with 82% of patients having UFR <10 mL/hour/kg.22 The cohort also exhibited a statistically significant decline in antihypertensive use, averaging 1.51 agents per day at baseline and 0.91 agents per day after 12 months of frequent therapy (p<0.001).22 The number of participants requiring no antihypertensive medication increased from 27% to 42% (p<0.001), and the number requiring at least two antihypertensive agents concurrently decreased from 43% to 25% (p<0.001).22
In a subset of 86 patients dialysing at home for ≥5 sessions per week, under the care of the Wessex Kidney Centre, Portsmouth, UK, 75% reported immediate recovery after dialysis. Across these 7,600 consecutive sessions on frequent therapy, a greater change in intradialytic systolic blood pressure was associated with increased number of symptoms reported (p<0.0001) and longer post-treatment recovery time (p<0.0001). Maintaining stable blood pressure reduces the likelihood of symptoms on dialysis, leading to fewer early sign-offs and less perpetuation of chronic fluid overload.15
An ageing multicomorbid dialysis population has resulted in a patient cohort that is, on average, more susceptible to fluid overload and haemodynamic instability than it was in the past.23,24 These patients are extremely sensitive to fluid shifts and are at a high risk of hospitalisation due to recurrent episodes of fluid overload, particularly after a 2-day gap in therapy.2 Furthermore, their haemodynamic instability compounds the difficulty in removing fluid.25 Providing patients with shorter, more frequent haemodialysis sessions and minimising UFR has been shown, in many cases, to engender fewer symptoms on dialysis, stabilise blood pressure, improve fluid balance, and reduce the risk of hospitalisation.21,22 The above data suggest that by directly addressing chronic fluid overload, frequent haemodialysis can meaningfully improve both cardiovascular outcomes and quality of life.
Frequent Home Haemodialysis in Complex Patients
Doctor Maxence Ficheux
Managing patients who are either pregnant or obese is uniquely challenging. Each of these patient types requires more volume clearance than the typical haemodialysis patient and presents unique physiological challenges and potentially poor health outcomes.26,27 More frequent haemodialysis in the home setting may improve outcomes in both pregnant patients and obese patients, as well as provide individualised and convenient therapy for these complex cases.
Women on haemodialysis are likely to experience decreased fertility due to both sexual dysfunction and reproductive hormonal dysfunction, including amenorrhea.28,29 Specifically, lack of oestradiol-stimulated cyclic luteinising hormone secretion leads to ovarian failure (i.e., anovulation), and an elevated prolactin concentration contributes to abnormal uterine bleeding.30 These physiological obstacles have historically contributed to a low likelihood of successful pregnancy. Survey data from multiple continents have shown few pregnancies on dialysis and a <50% probability of live birth among pregnant patients.31-33
Intensive haemodialysis may help improve poor pregnancy outcomes. One meta-analysis that assessed dialysis schedules and pregnancy outcomes reported a negative correlation between weekly haemodialysis duration and the percentage of pregnancies ending in preterm (i.e., <37 weeks) delivery (R2=0.22; p=0.044).34 The same meta-analysis also reported negative correlations between the percentage of small for gestational age infants and both weekly haemodialysis duration (R2=0.54; p=0.017) and weekly haemodialysis frequency (R2=0.84; p=0.003).34 Recent survey results have suggested that although prenatal complications and preterm delivery are still frequent, neonatal survival has improved in the last 30 years, which may be linked to the rise of more frequent and longer haemodialysis sessions.35
Obesity, which is prevalent in approximately 1 in 4 dialysis patients in both the USA and France, is a common challenge.36,37 Patients with a BMI >30 kg/m2 require more volume clearance than patients with a normal BMI.27 Frequent haemodialysis in the home setting may address this challenge. Thirty-three (18%) patients in the KIHDNEy cohort had a BMI ≥30 kg/m2.22 One year after initiating haemodialysis for ≥5 sessions per week, these patients maintained a mean standardised Kt/V of 2.53 (standard deviation: 0.31) and mean UFR of 4.49 mL/hour/kg. Concurrently, mean serum phosphorus among these patients decreased by 4%, despite a 24% reduction in mean phosphate binder pill count. Furthermore, the use of antihypertensive agents declined by 41%.22
These data, along with multiple case studies, provide compelling evidence that frequent home haemodialysis with low-volume dialysate not only maintains adequate small solute clearance, but also leads to improved outcomes and provides patients with a convenient and individualised therapy. In a pregnant patient, increased frequency and duration of haemodialysis may increase the likelihood of live birth and permit the mother and neonate to return home more rapidly. In obese patients, increased frequency and duration of haemodialysis may lessen the need for oral medications and lower the incidence of dialysis-related side effects. Frequent home haemodialysis represents a practical and efficient method of therapy for complex patients and gives new hope to end-stage renal disease patients who wish to conceive but are unable to wait for a transplant.
Frequent Home Haemodialysis as Palliative Care
Doctor Sunita Nair
Since the first successful dialysis treatment in 1945, the goal of dialysis therapy has shifted from saving lives to sustaining lives, and subsequently to rehabilitation, with more frequent haemodialysis prescribed to improve health-related quality of life. However, patients still experience a high burden of symptoms, including pain, itching, lethargy, and restless legs. Dialysis patients also have substantial functional limitations, which place a heavy burden on family members and care partners.
The current dialysis delivery system focusses on achieving fluid volume and biochemical targets, aiming to reduce cardiovascular risk. When these targets can no longer be achieved, the common practice is to withdraw dialysis and assist patients embarking on end-of-life journeys. However, some dialysis patients with significant symptom burden and coexisting terminal illness may wish to continue dialysis. These patients’ goals do not align well with the current interpretation of palliative care, which is offered only to patients with a predictably short life expectancy and a desire to forgo life-prolonging treatment. In the UK, for instance, patients are expected to withdraw from dialysis before entering hospice care. A new palliative dialysis care model may address these currently unmet needs.
Merging renal care and palliative care is a dynamic process aimed at offering specialised care for people with chronic or serious illnesses. This care model may be appropriate at any age and at any stage in a serious illness and can be provided alongside curative and life-prolonging treatment. Comprehensive palliative care in patients with end-stage renal disease focusses on providing patients with symptom relief, supporting decision-making, and aligning treatment plans with patients’ goals. By adopting a multidisciplinary approach between the renal and palliative care teams, the model provides truly holistic care by integrating psychosocial and spiritual needs with medical needs, and co-ordinating both medical and social services, thereby providing an extra layer of support for patients and their families.
Frequent haemodialysis in the home setting is arguably the most straightforward method of implementing this new paradigm of palliative dialysis care. Frequent therapy has been shown to reduce symptom burden, reduce morbidity, and improve quality of life, particularly in patients with multiple comorbidities.38 Home haemodialysis also allows patients to dialyse in their preferred setting, which is especially important during a vulnerable time in life. The NxStage System One enables dialysis delivery in both home and hospice settings without a requirement of costly remodelling of the site. Furthermore, this modality requires less training time, is financially viable, and allows ongoing palliative input through its monitoring system.39
This paradigm shift in providing palliative dialysis to patients with coexisting terminal illness resets the treatment goals of dialysis, focussing almost entirely on controlling symptoms and individualising treatment targets. Palliative care should be part of the continuum of holistic, quality care for renal patients. In this way, we will continue to extend the dialysis paradigm from one that rehabilitates patients when life expectancy is long to one that comforts patients when life expectancy is short.







