BACKGROUND
Motor disability has a high prevalence and dramatic effects on daily life activity of multiple sclerosis (MS) patients. Several rehabilitative strategies are currently available to treat these patients but often their efficacy is suboptimal and their benefits are generally not long-lasting. In patients with neurological diseases, action observation therapy (AOT) has been proposed as an effective rehabilitative intervention for regaining motor function.1,2 AOT is based on visual stimulation and it is thought to act through the function of the mirror neuron system, an observation-execution matching system.3-6 In MS, functional MRI (fMRI) techniques provide valuable measures to monitor disease evolution and treatment effects and might increase our understanding of the mechanisms responsible for a favourable clinical outcome following rehabilitation. Recently, a novel fMRI analysis method has been introduced. Such a method allows a time-varying (dynamic) assessment of resting state (RS) functional connectivity (FC), thus capturing reoccurring patterns of interaction among intrinsic functional networks at rest.7 In this present study, AOT was applied in right (R)-handed MS patients with motor impairment of their R hand to assess: 1) whether this strategy may lead to a clinical improvement of motor deficits; 2) changes over time of dynamic RS FC following AOT; 3) the correlations between RS FC changes and improvement at functional motor scales.
METHODS
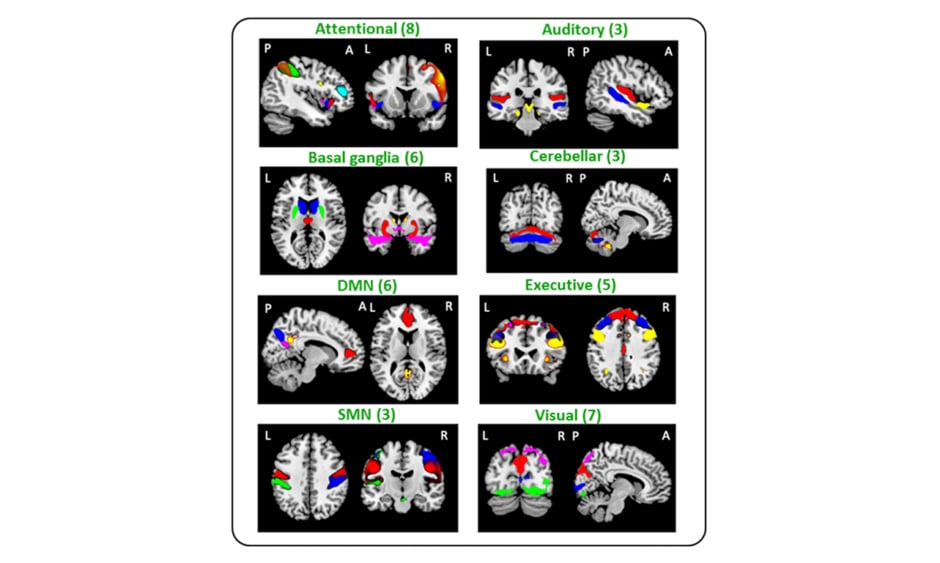
Eighty-seven R-handed subjects were randomised into two experimental-groups (healthy control (HC)-AOT=23; MS-AOT=20) and 2 control (C) groups (HC-C=23; MS-C=21). The groups underwent 2-weeks training of ten 45-minutes sessions. AOT-groups watched three motor-task videos alternated by their execution; C-groups performed the same tasks watching landscapes-videos. At baseline (t0) and after 2 weeks, RS fMRI and clinical evaluations were performed, including Nine-hole peg test, finger tapping (FT), Jamar and Pinch dynamometers, the Expanded Disability Status Scale (EDSS) test, Paced Auditory Serial Addition Test, and Functional Independence Measure. Independent-component analysis identified 41 FC-networks assigned to the sensorimotor, default mode (DMN), attention, executive, visual, auditory, basal ganglia, and cerebellar networks (Figure 1). Between-group differences and dynamic RS FC changes over-time were evaluated using a dynamic approach, assessing FC on small temporal segments using sliding-windows, and grouping FC correlation matrices into recurrent FC-states. Correlations between clinical and MRI-measures were also assessed.
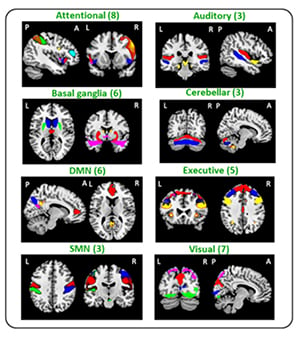
Figure 1: Composite map of the 41 identified independent components (IC) in all study subjects.
IC were sorted into eight subcategories (sensorimotor, default-mode, attentional, executive, visual, auditory, basal ganglia, and cerebellar networks). A: anterior; DMN: default mode; L: left; P: posterior; R: right; SMN: sensorimotor.
RESULTS
At t0, no significant differences were found in the comparison between the AOT and the respective control groups in both HC and MS. Decreased dynamic RS FC was detected in MS patients versus HC, mainly between the basal ganglia and cerebellar networks/DMN (p range: <0.001–0.01). Increased dynamic RS FC among visual, executive, and attention networks was also detected (p range: <0.001–0.008).
Dynamic RS FC significantly increased after two weeks of training in both MS groups, with more evident effects in MS-AOT than in MS-C, involving sensorimotor, visual, basal ganglia, DMN, and attention networks (p range: 0.001–0.009). Conversely, both HC groups showed a decrease of dynamic RS FC at Week 2 versus t0, mainly for the sensorimotor, basal ganglia, cerebellar, and attention networks in HC-AOT, and DMN and attention network in HC-C (p range: <0.001–0.01) .
Both in MS-AOT and MS-C, significant correlations were found between dynamic RS FC modifications of sensorimotor, DMN, and attentional networks, and improvement of motor performance at FT and Pinch (r range: 0.49–0.77, p<0.05).
CONCLUSIONS
A 10-day manual dexterity training with AOT modulated dynamic RS FC of several functional networks, with stronger effects in the MS-AOT than in the MS-C group. Dynamic RS FC modifications correlated with concomitant motor performance improvements, suggesting an adaptive role of the detected RS FC increase. These findings might improve the understanding of the functional substrates underlying motor deficit recovery in MS patients and contribute to developing individualised treatment strategies.