BACKGROUND AND AIMS
Brain tumour-related epilepsy (BTRE) is associated with diffuse lower-grade gliomas (DLGG) in up to 70–90% of cases.1 Several factors may affect seizure outcome and control, some of which are related to tumour characteristics (such as tumour location, histotype, molecular features, tumour growth, and progression), while others are the consequences of tumour treatment (extent of surgery, radiation therapy, chemotherapy, and the use of target agents).2-6 The authors’ study aimed to describe the natural history of BTRE in a real-life cohort of patients with DLGGs, to correlate the presence of seizures to tumour and patient characteristics, and to assess the impact of surgery and adjuvant treatments on seizure control.
MATERIALS AND METHODS
The authors retrospectively collected clinical data of patients with DLGGs, aged ≥18 years of age, with a history of BTRE. They retained information about seizure freedom after surgery, adjuvant treatments, and recurrence. The authors’ main endpoints were seizure freedom after surgery, adjuvant treatments, and recurrence.
RESULTS
This study included 280 patients (median age: 40) with DLGGs who were diagnosed between 1988 and 2021. Out of the 280 patients, 106 (54.9%) had oligodendrogliomas IDH-mutant 1p19q-codeleted, 40 (20.7%) had astrocytomas IDH-mutant, and 47 (24.4%) had IDH-wildtype. There were 199 (71.1%) patients with Grade 2 tumours and 81 (28.9%) patients with Grade 3 tumours. Gross total resection (GTR) accounted for 117 (41.8%) cases.
Seizure freedom at 30 days after surgery was seen in 144 out of 280 (51.4%) patients, and in 97 out of 151 (64.2%) patients after 6 months after starting adjuvant treatment. One-hundred and thirty-four (59.6%) patients displayed seizures as a symptom of recurrence. Patients with seizure persistence after surgery were significantly more likely to be of a younger age and have oligodendroglial tumours, Grade 2 histology, temporal lobe location, and incomplete surgery.
Furthermore, seizure freedom after adjuvant treatment prevailed among patients displaying an objective Response Assessment in Neuro-oncology (RANO) response to adjuvant treatments (being achieved in 84.2% versus 59.4% patients with complete/partial response versus stable/progressive disease; p=0.040). In a multivariable model (Table 1), seizure freedom after surgery was positively related to age ≥40 (odds ratio [OR]: 1.027; p<0.001) and GTR (OR: 1.788; p=0.049), and negatively related to Grade 2 histology (OR: 0.301; p<0.001). Similarly, Grade 2 histology (OR: 0.232; p=0.011) and temporal lobe location were negative predictors of seizure freedom after adjuvant treatments (OR: 0.318; p=0.041). Previous radiotherapy significantly reduced the risk of seizures at recurrence (OR: 0.233; p=0.026).
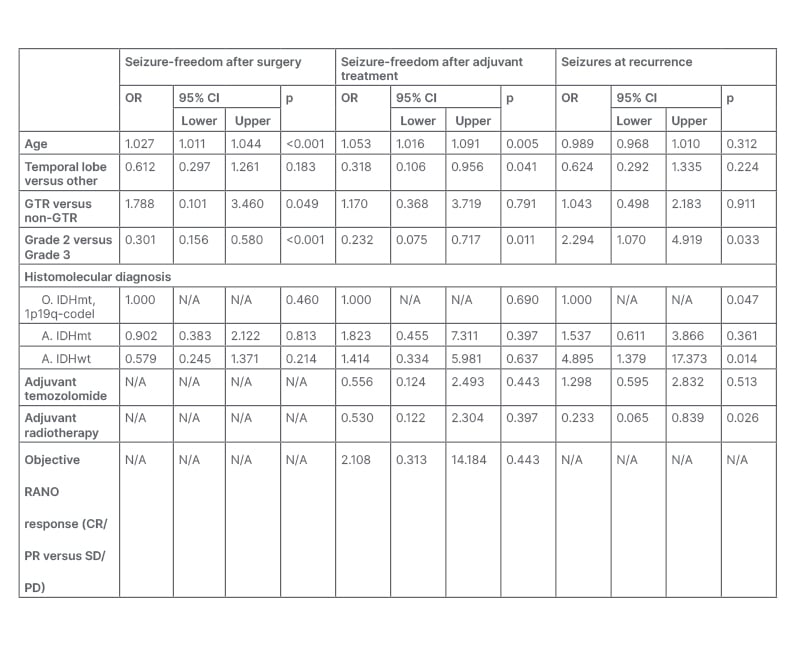
Table 1: Multivariable model of factors affecting seizure freedom after surgery, after adjuvant treatments, and at recurrence.
A: astrocytoma; codel, codeleted; CI: confidence interval; CR: complete response; GTR: gross total resection; IDH: isocitrate dehydrogenase; mt: mutant; O: oligodendrogliomas; OR: odds ratio; PD: progressive disease; PR: partial response; RANO: Response Assessment in Neuro-oncology; SD: stable disease; wt: wildtype.
CONCLUSION
In the authors’ study, younger age, Grade 2 histology, oligodendroglial phenotype, and temporal lobe involvement were baseline factors associated with a worse seizure control in patients with DLGGs. The extent of resection and adjuvant treatments affected seizure control at different levels: GTR correlated with a higher rate of seizure freedom after surgery; objective RANO responses to MRI (complete/partial response) prevailed among seizure-free patients after adjuvant treatments; and adjuvant radiotherapy was associated with a lower risk of seizures at recurrence. These data should be validated in larger prospective studies and the analysis should be extended to the new entities of the 2021 (World Health Organization) WHO Classification of brain tumours (astrocytomas IDH-mutant Grade 4 with CDKN2A/B homozygous deletion and astrocytomas IDH-wildtype with molecular features of glioblastoma).







