INTRODUCTION
Because of the rarity and high mortality of gallbladder cancer (GBC), the best adjuvant modality of radically resected GBC is not well established. Before the publication of the BILCAP study (adjuvant capecitabine versus observation in biliary tumours), the authors were practicing chemoradiation as adjuvant therapy. Over the past few years, the authors have offered chemotherapy (CT) as an adjuvant for moderate risk patients. Hence, the authors audited their records over a decade (2007–2017) to evaluate the change in outcomes with the introduction of CT (cisplatin-gemcitabine combination).
MATERIALS AND METHODS
In the period 2007–2012, only concurrent chemoradiation therapy (CTRT; n=40) was practised. Since 2013, low-risk patients were kept on observation (R0, T2, T3, N0), moderate-risk patients (R0, T2, T3 and N0, N1) received CT, and high-risk patients (R1, N2, T3, T4, lymphovascular invasion, perineural invasion) received CTRT. The lack of concrete guidelines for adjuvant therapy according to risk stratification and treatment offered according to physician discretion resulted in an overlap of treatment modalities (CT versus CTRT). Univariate and multivariate analysis was performed to ascertain the effect of different treatment modalities on prognostic factors and outcomes (overall survival [OS]) using SPSS (v.20).
RESULTS
The median age of patients (N=142) was 50 years (interquartile range: 42–58 years). At a median follow-up of 50 months, the median OS of all patients was 34 months. Between 2007–2012 and 2013–2017 period, the gain in median OS was 6 months (42 to 48 months; p= not significant) and the median disease-free survival improved by 3 months (30 to 33 months; p= not significant). The median OS was not reached versus 46 months versus 32 months with CT, CTRT, and observation, respectively (p=0.24). On univariate analysis, the median OS of patients <50 years was 48 months versus 42 months for >51 (p=0.29), females had better OS (50 months versus 26 months; p=0.07), those with comorbidities had worse outcomes (26 months versus 48 months; p=0.29). T2 patients had the best OS (72 months versus 40 months [T3] versus 16 months [T4]; p=0.13), node negative had better OS than node positive (72 months versus 40 months; p=0.08). The effect of various adjuvant therapy modalities on OS based on the prognostic factors is given in Table 1. The superior efficacy of CT was irrespective of the nodal status. The higher efficacy of CT was evident in T2 disease but not T3 disease. CTRT showed marked benefit in younger patients, females, and those with T3 disease. On multivariate analysis, the hazard ratio (HR) of various prognostic factors influencing OS were resection status (HR: 2.49; p=0.00), male sex (HR: 1.3; p=0.25), T status (HR: 2.1; p=0.15), and nodal status (HR: 1.3; p=0.2)
Locoregional recurrence rate was 37% with observation, 13% with CT, and 18% with CTRT, while respective distant metastases rate were 18%, 27%, and 20%.
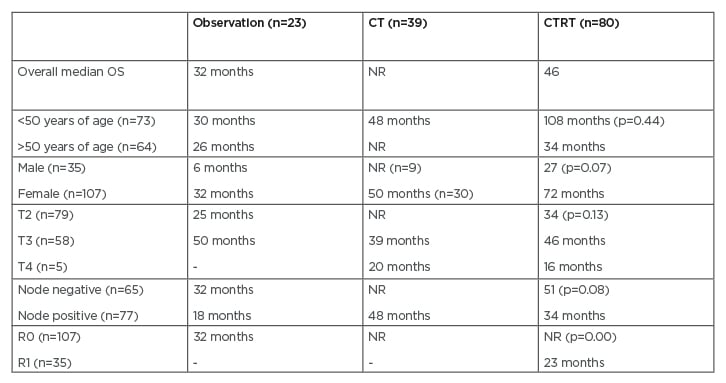
Table 1: The effect of various adjuvant therapy modalities on overall survival.
CT: chemotherapy; CTRT: chemoradiation therapy; NR: not reached.
CONCLUSION
The outcomes of postoperative GBC have improved in terms of OS and DFS over the years. All GBC patients should receive adjuvant therapy after radical surgery. Young age, female sex, early T stage, and node negative status are good prognostic factors for OS. CT should be the standard of care as adjuvant therapy for low-risk patients. CTRT should be used in patients with high-risk features, such as R1 resection status, lymphovascular invasion, and perineural invasion. These findings need to be validated in a larger database.






