BACKGROUND AND AIMS
Vacuum-assisted biopsy (VAB) is performed when a breast lesion appears suspicious at imaging examinations. Although several different modalities can guide the biopsy, some lesions can only be visualised by imaging modalities that can depict tumour neo-angiogenesis.1,2 For this type of lesion, MRI is the most sensitive modality when lesions are occult at second-look ultrasound or mammography.3 However, capacity is often insufficient, and waiting lists tend to be long. In this perspective, contrast-enhanced dedicated breast CT (bCT) could be a promising, faster, and less expensive alternative to biopsy lesions that are only detected by contrast enhancement. bCT provides reconstructed, isotropic, 3D images of the breast, with high spatial and contrast resolution. When coupled with the administration of intravenous contrast agent, the modality allows for the visualisation of tumour angiogenesis. Some of the commercially available bCT scanners can be equipped with a biopsy bracket to perform bCT-guided vacuum-assisted biopsies. The 3D guidance procedure allows for the localisation, targeting, and performance of biopsies.
While two previous studies, from 2013 and 2017, showed promising results with bCT-guided VAB in non-enhancing lesions,4 to the authors’ knowledge, the potential of bCT-guided VAB in contrast-enhancing lesions has never been investigated.
The aim of this study was to evaluate the feasibility, accuracy, success rate, and intervention time of bCT-guided VAB for enhancing lesions in breast phantoms.
MATERIALS AND METHODS
bCT-guided VAB was performed in four different types of phantoms (chicken breast, kiwi fruit, banana, and bread) to determine the targeting accuracy in breasts of different consistency. Iodinated contrast-enhanced olives (N=10) were inserted in the phantoms to simulate enhancing mass lesions. Biopsies were performed using a dedicated bCT (CBCT 1000, Koning Corp., Norcross, USA) (Figure 1). Biopsies were performed with the 9G ATEC® Sapphire vacuum-assisted breast biopsy console (Hologic, Marlborough, Massachusetts, USA). Targeting was deemed accurate when the centre of the biopsy chamber was ≤5 mm from the predetermined target. The technical success rate (defined as olive in the sample), time for identification/targeting of lesions and tissue sampling, as well as total intervention times were documented.
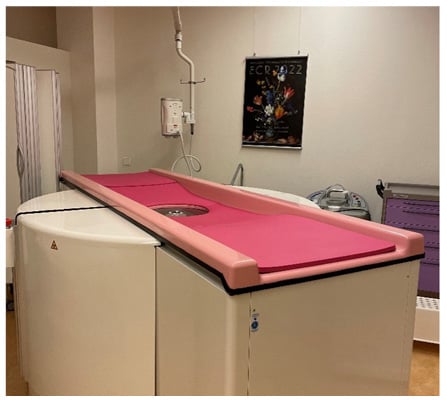
Figure 1: Contrast-enhanced breast CT.
RESULTS
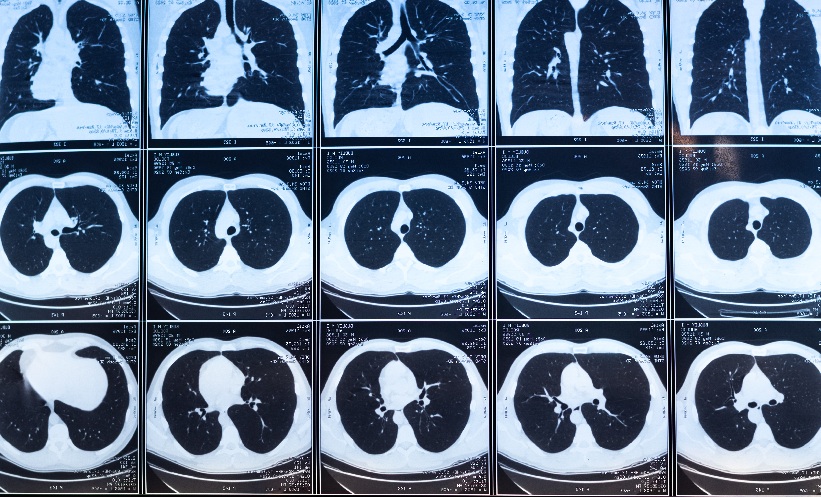
Ten 9G VABs were performed to biopsy 10 lesions. Technical success was achieved in all 10 lesions (100%) (Figure 2). During the biopsy on a kiwi phantom, needle repositioning was needed twice, as the distance to the target lesion was 7 mm and 6 mm in the first two attempts. The identification/targeting of lesions (Figure 3A) and tissue sampling took 11.5 min (10.2 min; 16.7 min) and 10.5 min (9 min; 12.5 min), respectively. The median (interquartile range) total intervention time was 22.5 min (19 min; 27.7 min). Twelve targeting attempts were needed, resulting in an accuracy of 90% (Figure 3B). For all 10 lesions biopsied, the location of the biopsy cavity was at the site of the lesion on the control scan, which was performed at the end of the procedure (Figure 3C). The median (interquartile range) size of the biopsy cavity was 14.5 mm (12.2 mm; 17.5 mm).
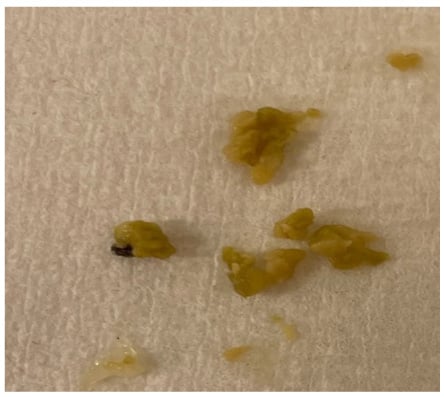
Figure 2: Olives in the sample after performing vacuum-assisted biopsy.
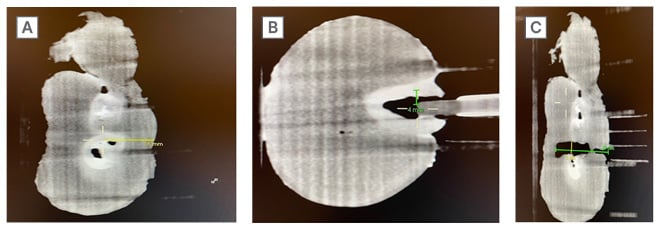
Figure 3: Different steps of performing a breast CT-guided vacuum-assisted biopsy.
A: Identification of the lesion on a chicken breast phantom.
B: Targeting of the lesion on a kiwi phantom.
C: Measurement of the lesion cavity at the control scan.
CONCLUSION
Success rate of bCT-guided VAB for enhancing lesions in breast phantoms was comparable to that of MRI-guided VABs reported in literature (100% versus 87–99%).5 The median total intervention time was substantially shorter compared to that reported in literature for MRI-guided VABs (22 minutes versus 72 minutes).6 However, these breast phantoms may not necessarily always simulate real patients, and therefore studies in patients are needed.