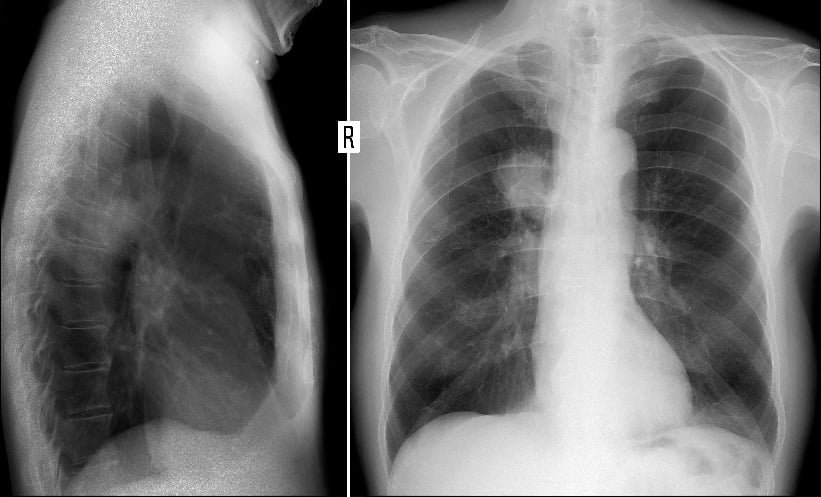
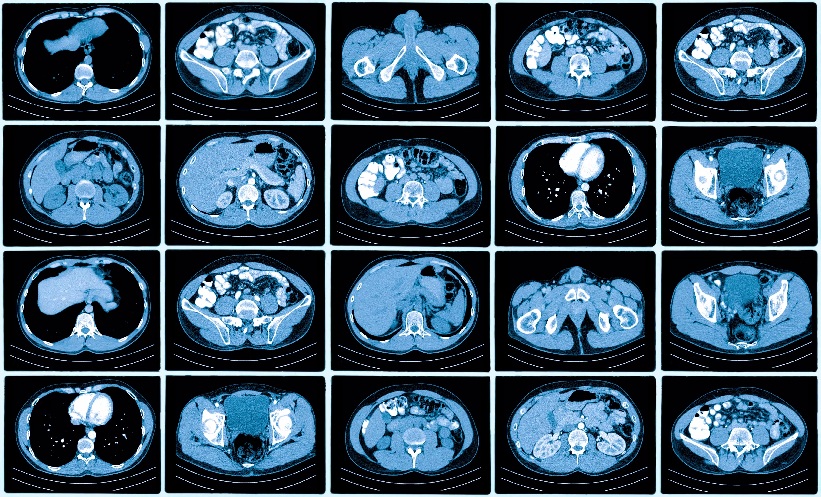
ACCORDING to a new study from the Karolinska Institutet (KI), Solna, Sweden, combining a new blood test with MRI could minimise the cost of prostate cancer (PCa) screening as well as the overdiagnosis of low-risk cancer. Conventional biopsies combined with prostate-specific antigen (PSA) tests have traditionally resulted in the detection of several smaller low-risk tumours. However, MRI has proven to reduce the extent of overdiagnosis.
A STHLM3MRI trial from the researchers at the KI has demonstrated that a new blood test called Stockholm3 could significantly reduce the number of MRIs by one-third for a screening episode. This research group currently reports that merging the blood test and MRI is more cost-effective in Sweden compared with both no screening and the traditional PSA test with MRI screening. A PSA test combined with MRI is categorised as expensive per cost per quality-adjusted life-years gained. Furthermore, the study forecast a reduced rate of 7–9% PCa-related deaths over a lifetime when MRI screening is combined with either PSA testing or Stockholm3 compared with no screening whatsoever.
“This new combination with Stockholm3 can save healthcare resources and reduce societal costs while maintaining the health benefits from early detection of PCa. This presents an interesting option for PCa screening in Sweden,” says first author Shuang Hao, Department of Medical Epidemiology and Biostatistics, KI, Solna, Sweden.
Various regions in Sweden have planned and implemented trial projects for organised PCa testing with regional cancer centres and intend to explore the utilisation of different strategies for PCa screening. Although the health economic analysis is specific for Sweden, the simulation model utilised is open source and the Stockholm3 test is available for clinical use in Norway, Denmark, Finland, Spain, and Germany, and will be made available other European countries this year.