BACKGROUND AND AIMS
Menopause hormone therapy (MHT) remains the most effective treatment for vasomotor symptoms and other manifestations of the climacteric. MHT also improves bone metabolism and urogenital health. However, its cardiovascular safety profile remains debated, particularly when therapy is initiated later in the menopausal transition.1,2
The ‘timing hypothesis’ proposes that cardiovascular outcomes associated with MHT depend on the interval between menopause onset and initiation of therapy.3 According to this hypothesis, initiation closer to menopause may confer cardiovascular protection, whereas later initiation may be associated with increased cardiovascular risk.4 Following publication of the Women’s Health Initiative (WHI) trials, clinical practice shifted toward a more cautious approach to hormone therapy, particularly in women aged ≥60 years, or ≥10 years since menopause onset.3,4
Current menopause society guidelines recommend individualised decision-making in these patients, although the evidence supporting these recommendations remains limited.5 The study presented at the International Society of Gynecological Endocrinology (ISGE) Congress aimed to evaluate cardiovascular outcomes associated with late initiation of MHT through a systematic review of RCTs.1
MATERIALS AND METHODS
A systematic review of RCTs was conducted to evaluate cardiovascular outcomes associated with late initiation of MHT. A comprehensive literature search was performed across PubMed, Embase, SciELO, and the Cochrane Library from database inception–November 2023.
RCTs evaluating systemic hormone therapy in healthy women aged ≥60 years or those ≥10 years post-menopause were included. Eligible interventions comprised oestrogen-only therapy, combined oestrogen–progestogen therapy, and tibolone.
Primary cardiovascular outcomes included coronary heart disease events, stroke, venous thromboembolism, and cardiovascular mortality. Study selection, data extraction, and risk-of-bias assessment were performed independently and in duplicate following PRISMA methodology and using the Cochrane Risk of Bias tool (RoB2) for randomised trials.6,7
RESULTS
Nine RCTs involving 36,051 participants met the inclusion criteria.1 Six studies were derived from analyses of the WHI trials, while three were independent randomised studies evaluating different hormone therapy regimens.3,4 The mean follow-up duration across studies was 7.2 years. The main characteristics and cardiovascular findings of the randomised trials included in this review are summarised in Table 1.
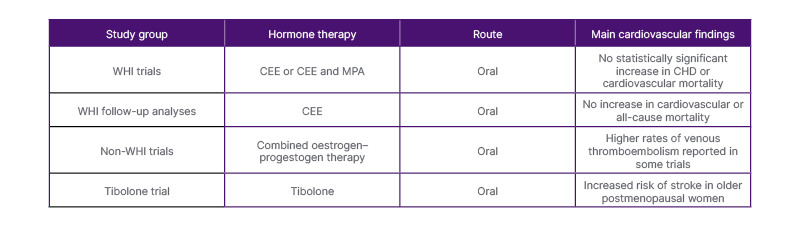
Table 1: Summary of randomised trials evaluating cardiovascular outcomes of late initiation of MHT.
CEE: conjugated equine oestrogen; CHD: coronary heart disease; MHT: menopause hormone treatment; MPA: medroxyprogesterone acetate; WHI: Women’s Health Initiative.
Across WHI-derived analyses, oestrogen-only therapy and combined oestrogen–progestogen therapy did not demonstrate a statistically significant increase in cardiovascular risk among women aged ≥60 years.3,8 Hazard ratios for coronary heart disease, stroke, and cardiovascular mortality were generally comparable between hormone therapy and placebo groups. Long-term follow-up analyses of WHI cohorts also showed no increase in cardiovascular mortality associated with oestrogen therapy.8
However, findings from non-WHI trials showed some variation depending on the hormonal regimen evaluated. Tibolone therapy was associated with an increased risk of stroke in older postmenopausal women in one randomised trial.9 Additionally, the WISDOM trial reported increased rates of venous thromboembolism among women receiving combined oestrogen–progestogen therapy, while other cardiovascular outcomes did not reach statistical significance.8
CONCLUSION
Available randomised evidence suggests that late initiation of MHT is not consistently associated with a statistically significant increase in cardiovascular risk. Nevertheless, the evidence remains heterogeneous and largely derived from secondary analyses of WHI trials.3,8
Differences in hormone formulations, doses, treatment duration, and patient characteristics across studies may influence cardiovascular outcomes.5 Furthermore, all trials evaluated oral oestrogen regimens, leaving an important evidence gap regarding other routes of administration, such as transdermal therapy.2
Given the multifactorial nature of cardiovascular risk, clinical decision-making should incorporate individualised risk assessment, including baseline cardiovascular risk factors, rather than relying solely on time sincemenopause onset.
Current randomised evidence does not demonstrate a statistically significant increase in cardiovascular risk associated with initiating MHT in women aged ≥60 years, or ≥10 years post-menopause.1 However, the limited number of randomised trials and variability among hormone regimens highlight the need for further research evaluating contemporary hormone therapy formulations, lower doses, and alternative routes of administration.