BACKGROUND AND AIMS
Study Question
Following testing via endometrial microbiome metagenomic analysis (EMMA) and analysis of infectious chronic endometritis (ALICE) for patients with recurrent implantation failure (RIF), is there any positive impact identified after the suggested treatments?
Study Answer
The clinical pregnancy rate of the patients who underwent EMMA/ALICE testing was significantly higher than that of the patients who did not undergo testing.
What Is Already Known
Chronic endometritis is persistent endometrial inflammation, mainly caused by bacterial infections. Chronic endometritis is found in approximately 30% of women with infertility,1,2 and 60% of patients with RIF;3 pathogenic flora identification is the first step of treatment. Using next-generation sequencing technology, EMMA/ALICE testing can determine the composition of the endometrial microbiome by analysing bacterial 16S ribosomal RNA with a focus on Lactobacillus population. Lactobacillus-dominated microbiota (LDM, defined as >90% Lactobacillus species) in the endometrium was reported to be associated with favourable reproductive outcome, while patients with non-LDM (<90% Lactobacillus species) endometrium were found to have decreased rates of implantation, clinical pregnancy, and ongoing pregnancy.4
METHODS AND MATERIALS
Study Design, Size, and Duration
The prospective cohort study consisted of 158 females with RIF, defined as at least three previous failed in vitrofertilisation–embryo transfer (ET) attempts, from July 2018 to March 2020 at one infertility centre in Japan. EMMA/ALICE testing were suggested to all patients who had failed ET three or more times. The study group of 107 patients underwent EMMA/ALICE before additional transfer, while 51 patients with history of RIF continued with ET without these tests, to make up the
control group.
Participants/Materials, Setting, and Methods
During the patients’ luteal phase, endometrial biopsies were performed for EMMA/ALICE testing, and treatment was provided based on the results.
The primary outcome measure was the cumulative clinical pregnancy rate after two additional ET. Clinical pregnancy was defined by visualisation of a gestational sac. Statistical analysis was performed using unpaired t-test and chi-square contingency.
RESULTS
Patients’ demographic data, such as age, BMI, duration of infertility, and anti-Müllerian
hormone, were comparable between the groups. The mean numbers of implantation failure were also comparable between the groups (study group: 5.11±2.79 versus control group: 5.50±2.26). According to the results of EMMA, 52 patients (49%) with normal microbiota (i.e., LDM) did not receive probiotic treatment; the other 55 (51%) patients received probiotic therapies. In addition, among non-LDM patients, dysbiotic microbiota were detected in 23 patients (21%) by EMMA, and ALICE detected significant amounts of pathogenic bacteria in 9 patients (corresponding to 39% of the patients with abnormal EMMA). Those 23 patients received antibiotic therapy according to the detected bacteria, followed by probiotic treatments. The cumulative clinical pregnancy rate in the study group was significantly higher than in the control group (study group: 64.5% versus control group: 33.3%). The difference in the implantation rate and the ongoing pregnancy rate were statistically higher in the study group than in the control group (Table 1).
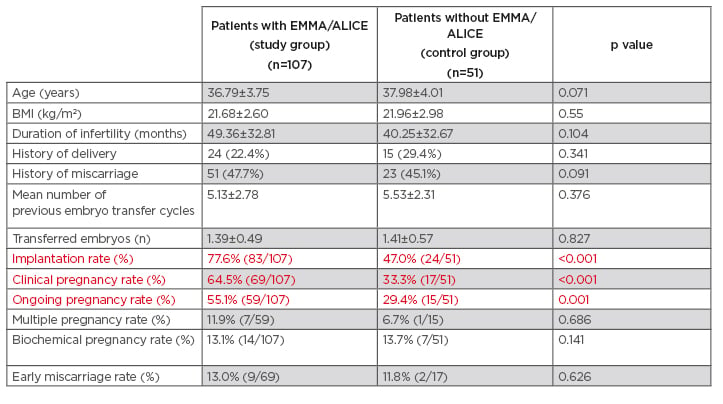
Table 1: Patient characteristics and pregnancy outcomes.
ALICE: Analysis of infectious chronic endometritis; EMMA: endometrial microbiome metagenomic analysis.
CONCLUSION
Limitation
The main limitation of the study was the lack of randomisation, although it was prospective.
Wider Implication
Next-generation sequencing technology revealed that >50% of the patients studied had dysbiotic microbiota. Personalised treatment recommendations based on the EMMA/ALICE results can improve in vitro fertilisation outcome of patients who have experienced RIF and/or repeated pregnancy loss. Moreover, broad-spectrum antibiotic treatments can be avoided, reducing the physical and economic burdens on patients.








