Meeting Summary
Menopause-related symptoms are common, varied, and often highly disruptive, significantly affecting women’s quality of life. The heterogeneous presentation of symptoms frequently necessitates tailored treatment approaches, yet important unmet needs remain for many women experiencing symptoms of menopause. Based on recent clinical trial evidence from the OASIS programme, dual neurokinin-targeted therapy (NKT) has been approved as a new therapeutic option for treatment of moderate-to-severe vasomotor symptoms (VMS; hot flashes, or night sweats if occurring at night) associated with menopause or caused by endocrine therapy (ET) for breast cancer. This article is based on the symposium, ‘Neurokinin-targeted therapy: a new approach to treating menopause symptoms’, delivered by four leading experts during the 40th Anniversary Congress of the International Society for Gynecological Endocrinology (ISGE) in Rome, Italy, in March 2026. The symposium, which featured a lively blend of panel discussions and presentations, was opened and chaired by Tommaso Simoncini, Obstetrics and Gynaecology, University of Pisa, Italy, and current President of the ISGE.
The Burden of Menopause
Millions of women enter menopause each year, often experiencing adverse effects such as VMS and sleep disturbances.1-3 VMS, including hot flashes or night sweats if the hot flashes occur at night, affect up to 80% of women, with as many as 40% reporting moderate-to-severe symptoms.2,3 VMS are strongly associated with reduced quality of life and changes in mood and cognitive function, particularly among women with moderate-to-severe symptoms.2,3 In addition, sleep disturbances, including poor sleep quality, insufficient sleep duration, obstructive sleep apnoea, restless legs syndrome, and insomnia, are reported by 69% of women, with prevalence increasing during the menopausal transition.2,4,5
Many women experiencing symptoms of the menopause remain untreated due to a culture of silence and a lack of acceptable treatments that meet their needs.6 There is a clear unmet need for effective treatments to address the substantial impact of menopause-associated symptoms, with Navarro noting that “the pathophysiology of menopause symptoms remains elusive, and the exact cause of hot flashes is still not fully understood.”
Currently, available treatments for menopause symptoms include hormone therapy, paroxetine and off-label antidepressants, and non-prescription/non-pharmacological approaches. While there are different options, they do not meet the needs of all women with symptoms of menopause. For example, hormone therapy is not appropriate for all women due to contraindications, risk factors, or adverse effects.7 It is also associated with high discontinuation rates,8 with perceived concerns about safety, meaning that women may be reluctant to start treatment, even after it has been prescribed.9 Paroxetine, the only selective serotonin reuptake inhibitor currently approved for VMS, is approved for this indication only in the USA and is sometimes used off-label in other countries.10 Other antidepressants used off-label for treatment of VMS are associated with tolerability issues as well as a lack of robust efficacy data.11 Use of non-prescription and non-pharmacological treatments, including phyto-oestrogen and other supplements, is also limited by conflicting data supporting efficacy and safety.12 “The substantial impact of menopause symptoms underscores the importance of providing effective treatments tailored to women’s individual needs and personal preferences,” commented Nappi.
One emerging approach in this area is NKT, which has shown clinical efficacy in the treatment of VMS associated with menopause. Elinzanetant, a dual neurokinin-1 (NK1) and neurokinin-3 (NK3) receptor antagonist, is currently approved in several regions including the EU and the USA, without a requirement or recommendation to track liver enzymes before or during treatment with elinzanetant in the EU but with a requirement for liver function testing prior to initiation and again after 3 months of treatment in the USA label.13,14 Fezolinetant, a mono NK3 receptor antagonist, is also approved in various regions for the treatment of moderate-to-severe VMS associated with menopause, and carries warnings regarding the risk of liver injury, including a requirement for liver function monitoring before initiation and during at least the first 3 months of treatment.15,16
KNDy Neurons and the Science Behind Menopause
Neurokinin Signalling in Menopause Pathophysiology and Treatment
Kisspeptin/neurokinin B/dynorphin (KNDy) neurons are a type of neuron found in the hypothalamus, and take their name from three of the neuropeptides that they release.17 KNDy neurons also release a fourth neuropeptide called substance P.17
During menopause, there is hyperactivity of KNDy neurons with upregulation of NK1 and NK3 receptors and their respective ligands Substance P (SP) and neurokinin B (NKB). NKB binds to NK3 receptors leading to dysregulation of the thermoregulatory center of the hypothalamus, resulting in hot flashes. SP binds to NK1 receptors and is involved in sleep disturbances.18 Peripheral NK1 receptors are also found on nerves associated with blood vessels in the skin and contribute to sweating and vasodilation as a cooling response to the increase in body temperature.19 KNDy neurons express oestrogen receptors, and their hyperactivity occurs due to a decline in oestrogenic activity as a result of natural or surgical menopause or ET (Figure 1).20-22
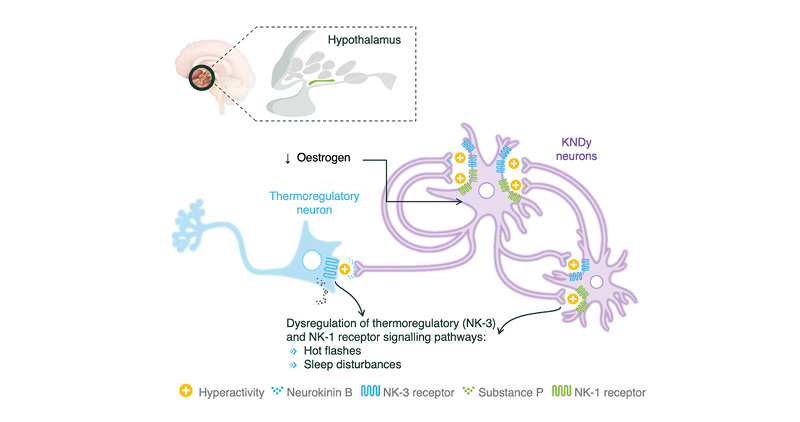
Figure 1: Role of KNDy neurons in the pathophysiology of menopausal vasomotor symptoms and sleep disturbances.17,21,23
KNDy: kisspeptin/neurokinin B/dynorphin; NK1: neurokinin-1; NK3: neurokinin-3.
This mechanistic understanding of KNDy neuron hyperactivity provides the biological rationale for targeting neurokinin signalling pathways in the management of menopause symptoms (Figure 2).18,19,23-25 NKTs act by antagonising NK1 and/or NK3 receptors expressed centrally within the hypothalamus and NK1 receptors located peripherally in the skin, thereby modulating the downstream effects of neurokinin B and substance P signalling.18,23-25 Through modulation of these central and peripheral neurokinin pathways, NKTs offer a non-hormonal approach to addressing both VMS, and additionally sleep disturbances, associated with menopause.18,23
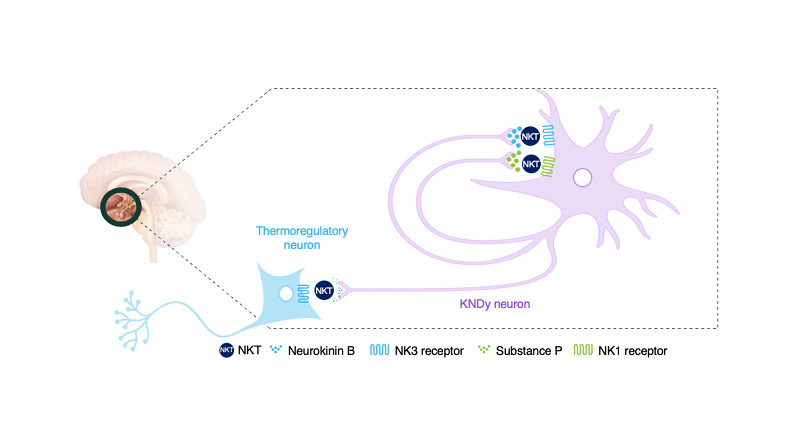
Figure 2: Mode of action of NKT in the regulation of body temperature.18-20,24,25
KNDy: kisspeptin/neurokinin B/dynorphin; NK: neurokinin; NKT: neurokinin-targeted therapy.
NKTs for the Treatment of VMS
Two NKTs are currently approved for treatment of VMS associated with menopause: elinzanetant and fezolinetant.13-16 The efficacy and safety of the non-hormonal dual NK1 and NK3 receptor antagonist, elinzanetant, were evaluated in four Phase III trials as part of the OASIS clinical development programme. OASIS-1 and OASIS-2 were pivotal multicentre, multi-country, double-blind, randomised, placebo-controlled studies of the efficacy and safety of elinzanetant taken orally, once daily for 26 weeks.18 OASIS-3 adopted the same study design over a period of 52 weeks, and focused primarily on safety,25 while OASIS-4 evaluated elinzanetant in a breast cancer population with VMS caused by ET.24 OASIS-1, -2, and -3 enrolled women aged 40–65 years (inclusive), with natural or surgical menopause, who were experiencing moderate-to-severe VMS and were seeking treatment for this condition.18,25 In OASIS-1 and OASIS-2, women were required to have at least 50 moderate-to-severe episodes of VMS per week,18 with no minimum number of VMS episodes per week specified for OASIS-3.25
Elinzanetant significantly and rapidly reduced the frequency of moderate-to-severe VMS compared with placebo, with reductions observed as early as Week 1 and maintained through Weeks 4 and 12 in OASIS-1 and -2.18 Reductions in severity were also observed.18 Numerical improvements in VMS frequency and severity were also maintained over 50 weeks in OASIS-3.
Fezolinetant, a mono NK3 receptor antagonist, was evaluated in two 12-week, placebo-controlled studies (SKYLIGHT 1 and SKYLIGHT 2) in which women treated with fezolinetant with ≥7–8 moderate-to-severe hot flashes per day (≥50–60 per week) at baseline were shown to have approximately 5–6 fewer VMS per day (36–43 per week) at Week 4 and approximately 6–7 fewer VMS per day (43–51 per week) at Week 12. Significant reductions in VMS severity were also observed at Week 12.26,27
NKTs for the Treatment of Sleep Disturbances
The impact of elinzanetant and fezolinetant was assessed in the OASIS and SKYLIGHT clinical programmes, respectively, based on mean change from baseline in the Patient-Reported Outcomes Measurement Information System Sleep Disturbance–Short Form 8b (PROMIS SD-SF 8b) total score.18,25-27 PROMIS SD-SF 8b assesses self-reported sleep disturbance during the prior 7 days, including perceptions of restless sleep, satisfaction with sleep, refreshing sleep, difficulties falling or staying asleep, amount of sleep, and overall sleep quality, with higher scores indicating more disturbed sleep.27
Across OASIS-1 and OASIS-2, elinzanetant produced significantly greater reductions in PROMIS SD-SF 8b total T-scores from baseline to Week 12 compared with placebo (both p<0.001). Reductions in sleep disturbances were sustained through Week 12 and beyond in OASIS-3, supporting durability of effect with continued treatment.18,25 The latest scientific research indicates that by targeting NK1 and/or NK3 receptors found centrally in the hypothalamus as well as NK1 receptors in the skin, elinzanetant helps bring balance back to temperature regulation and additionally sleep.18,19,23,24
Across the two SKYLIGHT trials, improvements in PROMIS SD-SF 8b scores were observed with fezolinetant; however, the magnitude and statistical significance of these effects varied by timepoint and study.26,27 In SKYLIGHT-1, observed improvements in patient-reported sleep disturbance with fezolinetant 45 mg versus placebo were not significant at Week 12,26 whereas in SKYLIGHT-2, statistically significant improvements were observed with the same dose at the same timepoint.27
Safety and Physiological Side Effects of NKT
In OASIS-1 and OASIS-2, the most frequent treatment-emergent adverse events (TEAE) observed during the 12-week placebo-controlled period were similar between groups. Headache was the most common TEAE, reported in 7% of treated patients in OASIS-1 and 9% in OASIS-2.18 Serious TEAEs were uncommon, and no cases of liver enzyme elevations meeting criteria for liver injury were identified.18 In the safety-focused OASIS-3 trial, elinzanetant was generally well tolerated over 1 year, with any TEAE occurring in 70.0% of women versus 61.1% with placebo. Headache, fatigue, and somnolence were more frequent with elinzanetant, but no serious TEAEs were considered treatment-related.25 Overall, elinzanetant demonstrated a favourable and consistent safety profile compared with placebo across the OASIS-1, OASIS-2, and OASIS-3 studies. No hepatotoxicity signal, Hy’s law cases, endometrial pathology, clinically relevant bone mineral density changes, or weight gain were observed.18,25
During the 12-week placebo-controlled periods of SKYLIGHT-1 and SKYLIGHT-2, the most frequent TEAEs with fezolinetant were generally comparable with placebo, with the most commonly reported TEAE in both trials being headache (11% of patients receiving fezolinetant 45 mg in SKYLIGHT-1 and 4% in SKYLIGHT-2).26,27 However, of greater concern, elevations in liver enzymes were observed in both trials, consistent with the warning that fezolinetant may rarely cause serious liver injury and therefore requires monitoring.15,16
In summary, NKTs may offer generally well-tolerated and efficacious treatment options for a broad population of women experiencing disruptive menopause symptoms. With respect to sleep disturbance, elinzanetant additionally showed consistent and statistically significant improvements versus placebo.18,25 These benefits were observed across a broad population of women, including those with natural and surgical menopause, supporting the role of NKTs as effective non-hormonal options in the management of disruptive menopause symptoms.
Managing Menopause Symptoms in Patients Undergoing ET due to HR+ Breast Cancer
Patients undergoing ET due to hormone receptor-positive (HR+) breast cancer represent a particularly important group of women as they may experience menopause symptoms, including a high prevalence of severe hot flashes, due to the cessation of menstrual cycles caused by the iatrogenic loss of ovaries,21,28 loss of ovarian follicular activity,21,28 suppression of oestrogen synthesis,29 and blockage of oestrogen receptors.21 Therapies that lead to oestrogen deprivation have the potential to affect multiple organ systems, including the reproductive, musculoskeletal, cardiovascular, and central nervous systems.30 Cano commented that “menopause symptoms caused by ET have a widespread impact on women’s quality of life.”
Pathophysiology of Menopause-Like Symptoms Caused by ET in HR+ Breast Cancer
When oestrogen levels decline during menopause, KNDy neurons within the hypothalamus undergo hypertrophy and become hyperactive.20-22,31 This reduction in oestrogen-mediated inhibition enhances neurokinin B signalling, which activates autonomic thermoregulatory pathways, resulting in cutaneous vasodilation and heat dissipation that manifest clinically as VMS, including hot flashes.20-22,31 In women receiving ET, similar mechanisms may contribute to symptom development. Tamoxifen binds to oestrogen receptors in the hypothalamus, thereby altering neurokinin B signalling, whereas aromatase inhibitors prevent the conversion of testosterone to oestrogen, further reducing circulating oestrogen levels.20-22,31 Together, these effects can exacerbate KNDy neuronal activation and thermoregulatory instability, contributing to menopause-like symptoms in this population.20-22,31
Non-hormonal Options for Women with VMS Caused by ET for Breast Cancer
Non-hormonal treatment options for VMS after cancer include cognitive behavioural therapy, hypnosis, and acupuncture. Cognitive behavioural therapy, which may be carried out either online or in person, has been shown to reduce VMS and improve mood, sleep, and sexual function.32 Similarly, hypnosis has been shown to reduce VMS and improve sleep,32 while acupuncture is more effective than no treatment (although this may be due to a placebo effect).33 However, these options have modest or inconsistent evidence of efficacy in women receiving ET, while adverse effects are common.32,33
Use of NKTs in the Treatment of VMS Caused by ET
Elinzanetant has been studied in the pivotal OASIS-4 trial in women with moderate-to-severe VMS caused by ET.24 This multicentre, multinational, double-blind, randomised, placebo-controlled study assessed the efficacy and safety of once-daily oral elinzanetant administered for 52 weeks, with an optional extension of up to 2 additional years. Eligible participants were women aged 18–70 years (inclusive) with moderate-to-severe VMS caused by adjuvant ET for HR+ breast cancer, or at high risk of developing HR+ disease, who experienced at least 35 moderate-to-severe VMS episodes per week.24
Elinzanetant significantly reduced the frequency of moderate-to-severe VMS compared with placebo in women experiencing VMS caused by ET, with the benefit observed as early as Week 1 and sustained throughout the 52-week study period (Figure 3).24 Elinzanetant also reduced the mean daily severity of VMS from baseline to Week 12, with the improvement sustained for 52 weeks.24 In terms of sleep disturbances, elinzanetant significantly reduced PROMIS SD-SF 8b scores from baseline–Week 12 versus placebo, with improvements in sleep disturbances sustained throughout the 52-week treatment period.24
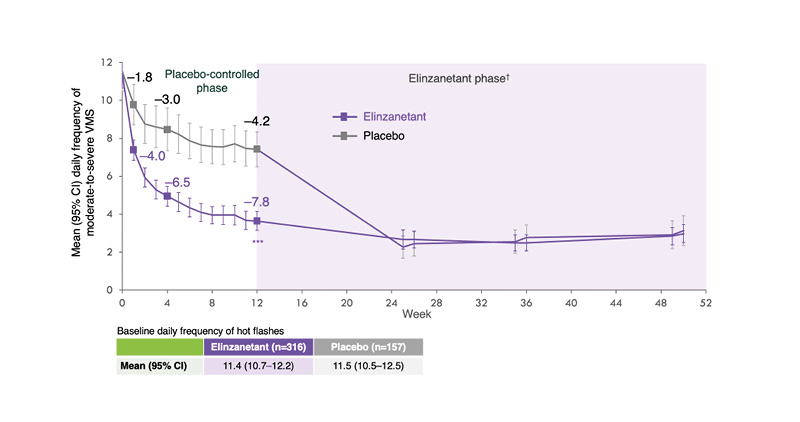
Figure 3: Change in mean daily frequency of moderate-to-severe vasomotor symptoms with elinzanetant (OASIS-4).19
***p<0.001
†Those who received placebo for the first 12 weeks switched to elinzanetant for the remainder of the trial.
Adapted from Cardoso F et al.19
ET: endocrine therapy; VMS: vasomotor symptoms.
The safety profile of elinzanetant in OASIS-4 was generally consistent with findings from OASIS-1, OASIS-2, and OASIS-3.18,24,25 Most TEAEs were mild or moderate in severity,24 while serious TEAEs were infrequent, occurring in 2.5% of participants receiving elinzanetant and 0.6% of those receiving placebo during Weeks 1–12.24 Importantly, the independent liver safety monitoring board concluded that there was no substantive hepatotoxicity signal associated with elinzanetant,22 consistent with data from OASIS-1, OASIS-2, and OASIS-3.18,25 Further long-term safety data are anticipated from the ongoing 2-year extension phase, in which 91.6% of participants who completed 52 weeks of treatment elected to continue receiving elinzanetant.24
As a result of the OASIS-4 data, the National Comprehensive Cancer Network (NCCN) Survivorship Guidelines were recently updated to include elinzanetant as a preferred non-hormonal pharmacologic option in the NKT category for the management of VMS in patients receiving ET for breast cancer.34 Published data on fezolinetant specifically in women with breast cancer are not yet available.
Conclusions
Menopause-related symptoms are common, multifactorial, and uniquely disruptive, with a substantial adverse impact on women’s quality of life. Due to the heterogeneity of the symptoms, they often require tailored treatment approaches that reflect the specific clinical profile, symptom burden, and underlying cause of menopause.
Despite the availability of hormonal and non-hormonal therapies, there remains an unmet need for effective and well-tolerated treatment options for women experiencing natural, surgical, or menopause symptoms caused by ET. In this context, clinical trial evidence from the OASIS programme is establishing dual NKT as a new option for addressing VMS with additional sleep benefits in women across these diverse menopause populations.
| Adverse events should be reported. Reporting forms and information for the EU can be found here. Adverse events should also be reported to Bayer via Safe Track. |