INTRODUCTION
Omalizumab is a recombinant humanised monoclonal anti-IgE antibody currently approved in the USA and Europe for treating inadequately controlled allergic asthma. Previous data have shown the high efficacy of omalizumab in severe asthma. However, factors that may affect response to therapy, relapse rates after drug discontinuation, and efficacy of retreatment remain unclear. This study aimed to evaluate the efficacy of retreatment on relapsed disease.
Methods
Patients who were treated with omalizumab at least 2 years ago at first treatment and were also retreated at least 1 year ago were included in the study. Patients with adverse effects causing discontinuation of first treatment were excluded. Social demographic data, laboratory findings, pulmonary function tests, asthma attack, hospitalisation due to asthma attack, systemic corticosteroid usage, asthma control test, and adverse effects during retreatment were recorded.
RESULTS
Twelve patients were included in the study. There were 8 female patients and the mean age of all patients was 57.4±11.9 years. The most popular reason for stopping omalizumab treatment was following the patient’s wishes (n=7). The retreatment dosage of omalizumab was found to be decreased (p>0.05). Mean duration of time from discontinuation of first treatment to retreatment was 12.2±9.7 months. Two patients did not respond to retreatment.
After retreatment, asthma control test was improved (16.2±2.7 versus 22.1±2.0 [p<0.05]). Emergency room visits, number of asthma attacks, hospitalisation due to asthma attack, and the number of patients needing systemic corticosteroid treatment were also decreased after retreatment (p<0.05) (Table 1).
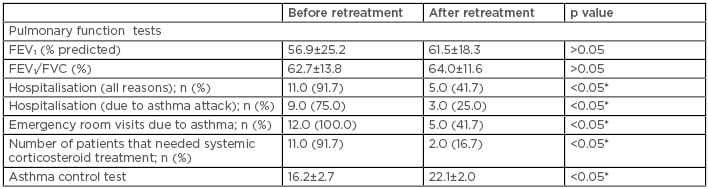
Table 1: Outcomes after omalizumab treatment.
*Significant p value.
FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity.
Two patients who were retreated with a lower dosage of omalizumab than at first treatment did not respond to retreatment with omalizumab, despite responding at first treatment. Regarding the adverse effects of treatment, there was no difference between first treatment and retreatment.
CONCLUSION
Omalizumab retreatment is effective and well-tolerated in patients with severe asthma who have benefited from initial omalizumab treatment.







