BACKGROUND AND AIMS
Diffuse tracheal wall thickening is often subtle, and carries a wide differential diagnosis. Radiographic features that can narrow the differential include posterior membrane sparing and calcification.1
MATERIALS AND METHODS
A 61-year-old male who had never smoked, with BMI of 24, presented to clinic for 3 years of progressive dyspnea (Modified Medical Research Council [mMRC]: Grade I) and dry cough. The cough was occasionally ‘barky’ in quality and was steroid-responsive. Physical exam was notable for mild expiratory wheezing, and rhinoscopy showed erythematous turbinates without structural abnormality and normal vocal cords/subglottis.
RESULTS
Pulmonary function tests demonstrated severe obstruction with forced expiratory volume in 1 second of 1.19 L (31% predicted), with positive bronchodilator response with an expiratory flow volume curve with biphasic appearance. Laboratories revealed elevation in C-reactive protein to 23.8 mg/L, with otherwise negative autoimmune and infectious testing. CT of the chest showed diffuse thickening of the anterior and lateral trachea sparing the posterior membrane with patchy calcification and tracheobronchomalacia involving the trachea to central bronchi. Further history was notable for sensorineural hearing loss that predated onset of the patient’s respiratory symptoms, and had resolved on audiology re-evaluation. Bronchoscopy revealed erythematous bronchial mucosa and mild tracheobronchomalacia, thought to be consistent with the appearance of relapsing polychondritis (RP). Using the modified Damiani criteria, the patient’s presentation was consistent with RP, given respiratory tract chondritis involving the trachea and bronchi on CT, sensorineural hearing loss thought likely due to RP, and steroid responsiveness.2 The patient was started on oral prednisone, and methotrexate was initiated, resulting in gradual improvement in cough and dyspnea. The patient remains under close follow-up to monitor for treatment response, as well as airway complications.
CONCLUSION
RP often presents as a diagnostic challenge, especially in those without typical features of chondritis or arthritis on presentation.3 Furthermore, a lack of specific serology, limitations in the role of biopsy, and lack of unified diagnostic criteria can complicate this diagnosis.4 Airway involvement occurs in around 50% of patients with RP, although rarely presents with isolated tracheobronchial disease.3,5 Several features on CT differentiate RP from other diffuse diseases in the trachea, including diffuse tracheal thickening sparing the posterior membrane, with or without patchy calcification as characterized in Figure 1.1,5
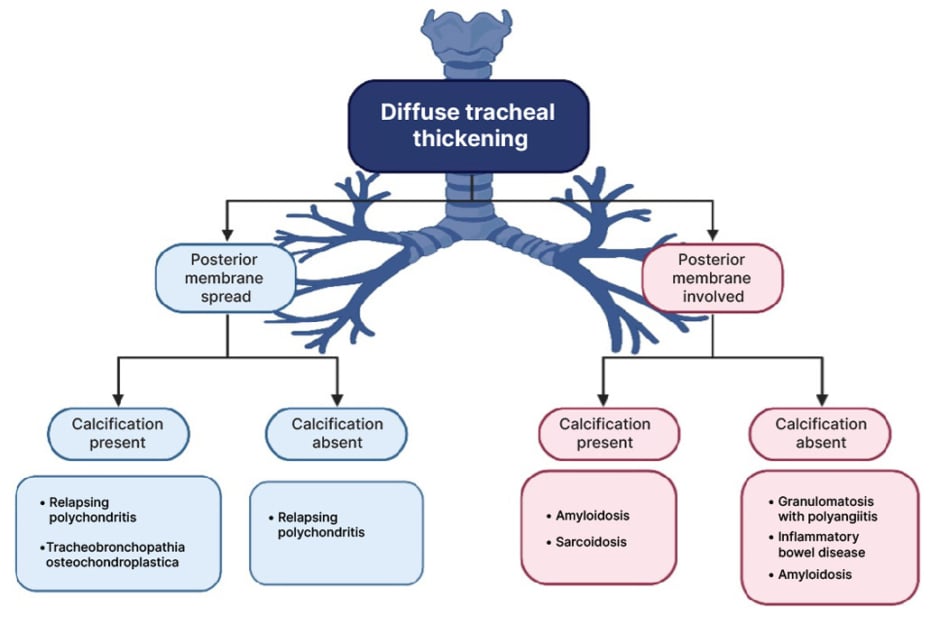
Figure 1: Differential diagnosis of diffuse tracheal thickening on imaging, using posterior membrane involvement and presence/absence of calcification as two key radiographic features.
Patients with RP and airway involvement represent a distinct clinical subset often associated with poorer prognosis with relapsing disease, as well as airway complications, including progressive stenosis, tracheobronchomalacia, and infection.1,6 The evidentiary basis for treatment of RP primarily based on case reports and case series, which is generally dictated the degree of organ involvement, although randomized trials are warranted.7 Treating patients with RP and airway involvement requires oral corticosteroids and as well as considering disease modifying anti-rheumatic drugs, oftentimes is the first line agent, as a substantial proportion of those with RP progress despite initial immunosuppression.4 In refractory cases of those with tracheobronchial disease, the use of biologics with most case series data supporting TNF-α inhibitors can be considered, although again, randomized data in this field are lacking.7,8 Patients with RP with respiratory manifestations not only require follow-up monitoring for treatment response and complications, but also for other evidence of concurrent autoimmune disease or hematologic malignancy, which occurs in a subset of those with RP.4,6







