Abstract
The prevalence of respiratory diseases, including asthma and chronic obstructive pulmonary disease, has increased in recent decades, placing a significant burden on healthcare systems and economies around the world. As these diseases are largely incurable, the aim of treatment is to control symptoms and improve quality of life. Aside from stopping smoking and reducing biomass fuel exposure, arguably the most effective strategy in the long-term management of chronic respiratory diseases is the prevention or control of respiratory infections via vaccines and antimicrobial agents. By preventing these infections or reducing exposure to some of the major risk factors, we can reduce further lung damage in these patients, thereby slowing disease progression. This review looks at maintaining long-term respiratory health in patients with asthma and chronic obstructive pulmonary disease, primarily through smoking cessation, reducing exposure to allergens and air pollutants, and infection control.
INTRODUCTION
The health, economic, and social burden of chronic respiratory diseases, including asthma and chronic obstructive pulmonary disease (COPD), is significant and increasing.1,2 In 2014, the average prevalence rate of asthma among adults across European Union (EU) countries was just over 6%, and for COPD it was 4%.3 It is estimated that COPD alone causes around 3 million deaths each year, and it is predicted to become the third biggest killer worldwide by 2030.4 To combat the increasing burden of these diseases, the Global Initiative for Asthma (GINA) was launched in 1993, followed by the Global Initiative for Chronic Obstructive Lung Disease (GOLD) in 1997. In recent reports (published in 2018),5,6 both GOLD and GINA advise three main approaches for preventing premature death from airway disease: early and accurate diagnosis, preventing progression, and prolonging survival.
While accurate diagnosis is important for the optimal management of respiratory disease, patients with COPD and asthma can present with similar symptoms (e.g., cough and dyspnoea), making differential diagnosis difficult. To reflect the overlap of the common disorders that cause airflow limitation, the diagnosis of asthma-COPD overlap (ACO) has been coined.6 Similarly, bronchiectasis and COPD share many pathophysiological characteristics and may coexist as an overlap syndrome similar to ACO.7 However, due to the paucity of randomised controlled trials (RCT) to guide treatment strategies, the GINA and GOLD guidelines currently recommend treating these overlapping diseases according to the dominant phenotype.
Smoking is the number one risk factor for developing COPD and is estimated to be responsible for ~80% of all COPD cases, although other factors (e.g., occupational exposures, a history of infections like tuberculosis, and genetic factors such as alpha-1 trypsin deficiency) can play a role.8 For asthma, the main risk factor in atopic individuals is exposure to allergens;9 for those with non-atopic asthma, smoking, air pollutants, occupational exposures, advanced age, lower socioeconomic status,9 and genetic factors may be involved.10 Therefore, policies aimed at reducing exposure to some of these risk factors (e.g., to tobacco smoke, allergens, and air pollutants) may reduce the incidence of COPD and asthma in future.
As there is currently no cure for COPD or asthma, the interventions recommended by GINA and GOLD aim to control symptoms and improve patients’ quality of life. The use of bronchodilators and anti-inflammatory drugs can improve symptoms such as shortness of breath. However, other interventions besides β2-agonists, antimuscarinics, and inhaled corticosteroids can reduce the symptomatic burden and prevent acute exacerbations. While the aetiology of these exacerbations can vary, the major causes are bacterial or viral infections.11,12 As the frequency and severity of acute exacerbations in patients with COPD and asthma is linked to disease progression (i.e., accelerated decline of lung function), reduced quality of life, and increased morbidity and mortality,13 infection control is a key consideration for maintaining long-term respiratory health. This review focusses on maintaining long-term respiratory health in patients with COPD or asthma through smoking cessation, reducing exposure to allergens and air pollutants, and infection control.
SMOKING CESSATION AND RESPIRATORY HEALTH
In 1998, the UK government published a white paper on tobacco, stating that smoking is the single greatest preventable cause of ill health.14 Smoking is associated with disease progression, increased mortality, and worsening of symptoms in both asthma and COPD.15 Asthmatic smokers have more severe symptoms and increased risk of exacerbations.16 In addition, asthmatic smokers have an elevated propensity to develop COPD or ACO.17 Smoking tobacco also directly increases the risk of subsequent respiratory infections, which again are associated with disease progression.18
As smoking is a modifiable risk factor, smoking cessation is strongly indicated by both GOLD and GINA as a clinical strategy to improve outcomes for patients with COPD or asthma.5,6 Smoking cessation in asthmatics improves lung function, symptoms, treatment outcomes, and quality of life.19,20 For COPD, smoking cessation is the only intervention that has been shown to slow the decline in the forced expiratory volume in one second (FEV1),21 reduce all-cause mortality,22 and improve all four core symptoms of COPD (chronic cough, chronic phlegm production, wheezing, and shortness of breath).23 The earlier the smoking cessation, the greater the impact on lung function.21 In addition, the use of e-cigarettes to help smokers with COPD reduce their cigarette consumption was recently found to lead to marked improvements in annual exacerbation rates.24 However, the use of e-cigarettes remains controversial, as their overall safety profile is not well defined.
Smoking cessation with pharmacological support ranks as one of the best value-for-money options among the available COPD interventions (£2,000 per quality-adjusted life year [QALY], according to the COPD value pyramid).25 Due to its cost-effectiveness, the European Respiratory Society (ERS) recommends that smoking cessation treatment be integrated into the management of a patient’s respiratory condition and pharmacological treatment should be included in combination with behavioural support and counselling.26 It is also strongly advised that those with asthma and COPD avoid environmental smoke exposure. This includes encouraging parents or carers of those with respiratory diseases not to smoke.5,6
AVOIDING ALLERGENS, BIOMASS FUEL SMOKE, AND OTHER AIR POLLUTANTS
Exposure to allergens and environmental pollutants (e.g., indoor and outdoor air pollution and occupational exposure) may also cause or exacerbate COPD or asthma.27 Indeed, it is increasingly becoming evident that indoor air pollution from biomass fuel exposure (i.e., the burning of wood, charcoal, etc., for cooking and heating) is an important risk factor for non-smoking-related COPD.28 To reduce the detrimental health effects of air pollutants, the Royal College of Physicians (RCP) and Royal College of Paediatrics and Child Health (RCPCH) presented a number of key recommendations in their report titled ‘Every breath we take: the lifelong impact of air pollution’.29 However, further research is required to determine the benefits of some of these strategies (e.g., the use of mixed fuel for cooking or heating) on respiratory diseases.28
The 2018 GINA report states that avoiding outdoor allergens (e.g., by closing windows and doors when pollen counts are high) may be beneficial in those with asthma (Table 1 [Evidence D]).6 While avoiding indoor allergens is not recommended as a general strategy in asthma, reducing exposure to mould can improve asthma symptoms (Table 1 [Evidence A]).6 In addition, the use of non-polluting heating and cooking sources (and adequate ventilation) is recommended for patients with asthma and COPD (Table 1 [Evidence B]).5,6 Finally, the most recent GOLD report advises that patients with COPD should avoid continued occupational exposure to potential irritants (Table 1 [Evidence D]).5
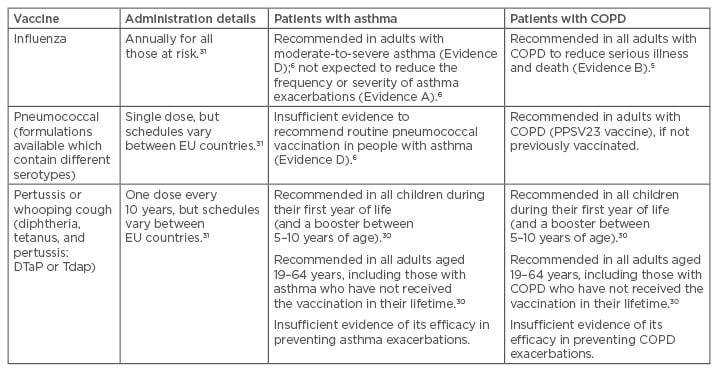
Table 1: Vaccination recommendations in patients with asthma or chronic obstructive pulmonary disease.
Evidence A: randomised clinical trials (rich body of data); Evidence B: randomised clinical trials (limited body of data); Evidence C: non-randomised trials (observational studies); Evidence D: panel consensus judgement.
COPD: chronic obstructive pulmonary disease; EU: European Union; FEV1: forced expiratory volume in one second.
VACCINATION AGAINST RESPIRATORY INFECTIONS
As viruses and certain bacteria have been shown to contribute to acute exacerbations in both asthma and COPD, vaccination is a key strategy to slow progression of these diseases.30 The current evidence for the use of vaccines in the management of COPD and asthma is summarised in Table 1.
Influenza Vaccine
There is solid evidence for a beneficial role of the seasonal influenza vaccine in patients with COPD. A systematic review found seasonal influenza vaccination reduces the number of acute exacerbations in those with COPD, presumably triggered by influenza virus infection.32 A retrospective cohort study in Spain also found that influenza vaccination reduced the risk of severe exacerbations in the year following immunisation, especially in those with severe COPD.2 Indeed, according to the COPD value pyramid, influenza vaccination is considered to be more cost-effective (£1,000 per QALY) than smoking cessation with pharmacological support (£2,000 per QALY) or the use of long-acting β2-agonists (£8,000 per QALY).25 However, a Cochrane review of the evidence from available RCT demonstrated no significant reduction in the frequency of asthma exacerbations following influenza vaccination.33 Indeed, according to the latest GINA report, seasonal influenza vaccination is not expected to reduce the frequency or severity of asthma exacerbations.6 Although there remains some debate regarding efficacy in reducing exacerbations, the European Centre for Disease Prevention and Control (ECDC) currently recommends annual influenza vaccination for all patients with COPD, as well as those with moderate-to-severe asthma.31
Haemophilus influenzae Vaccines
The nontypeable (non-capsulated) form of Haemophilus influenzae, a common bacterium in the nasopharynx, can cause considerable inflammation when present in the lower airways of patients with COPD, thereby worsening symptoms.34 While there are vaccines available against H. influenzae b that aim to prevent systemic infections (e.g., bacteraemia, meningitis) caused by the capsulated form of H. influenzae, unfortunately they have no activity against the non-capsulated forms of H. influenzae implicated in COPD exacerbations. An oral nontypeable H. influenzae (NTHi) vaccine has been developed; however, a Cochrane review demonstrated oral NTHi vaccination does not significantly reduce the number and severity of exacerbations in patients with recurrent exacerbations of chronic bronchitis or COPD.35 Nonetheless, there are new vaccines under development for NTHi (as reviewed by Murphy36) that may offer improved outcomes for those with COPD in future.
Pneumococcal Vaccines
Streptococcus pneumoniae (pneumococcus) colonisation occurs more frequently in the airways of those with COPD than healthy individuals, and colonisation is associated with a higher incidence of exacerbations.37 Those with asthma are also at increased risk of pneumococcal disease,38 which in turn increases the risk of wheezing.11 Therefore, injectable polyvalent pneumococcal vaccines may provide some protection against morbidity in those with COPD or asthma.
In addition to the current vaccination schedule (i.e., in all babies, and in those aged 19–64 years as required), pneumococcal vaccines are recommended for patients aged ≥65 years and for younger COPD patients with significant comorbidities.5 Clinical efficacy and recommendations for the use of pneumococcal vaccines in chronic respiratory disease (especially COPD) patients were recently reviewed by Froes et al.39 who concluded that: “[…] pneumococcal and influenza vaccinations can prevent community-acquired pneumonia and acute exacerbations in COPD patients, while pneumococcal vaccination early in the course of COPD could help maintain stable health status.”
As there is currently insufficient evidence to recommend routine pneumococcal vaccination in those with asthma (Table 1 [Evidence D]; panel consensus judgement),6,40 the UK Joint Committee on Vaccination and Immunisation (JCVI) currently only recommends immunisation for adults with COPD; asthma is not an indication, unless requiring continuous or frequent use of systemic steroids.41 However, next-generation vaccines against S. pneumoniae are being developed that could have a significant impact on respiratory health.42 Chalmers et al.43 recently called for more research into community-acquired pneumonia, which receives poor funding relative to its high disease burden, and for which contemporary data are required.
Pertussis Vaccine
Associations between Bordetella pertussis infection (whooping cough) and COPD have also been found; however, there is no association between B. pertussis infection and severity of COPD.44 In those with asthma, B. pertussis infection has been shown to lead to worsening of symptoms.30 Nonetheless, due to a paucity of clinical trials in the UK on the effectiveness of pertussis vaccination in patients with COPD or asthma, use of the vaccine in adults is currently only recommended for pregnant women to protect the newborn baby; specific guidelines for adults with chronic respiratory disease are lacking.30
Problems with Coverage
Although pertussis vaccination coverage is high across EU countries, vaccination rates for influenza and pneumococcal disease vary. For example, influenza vaccination coverage in clinical risk categories for all EU member states varies from 28.7–78.7%.31 In Spain, pneumococcal vaccination coverage was found to be less than acceptable in COPD patients aged >40 years (67.5% men and 60.4% women).45 The low vaccination rates may be due to a lack of awareness of infection-associated complications, anti-vaccination campaigns,46 and other factors (such as the recent ruling by the Court of Justice of the European Union [CJEU] in a compensation case, in which a man claimed that a vaccination for hepatitis B caused his multiple sclerosis).47
Some propose that vaccination will be most cost-effective in those that are at high-risk; indeed, a model was recently developed that can predict the risk of exacerbations in COPD and could be used to identify those who would most likely benefit from vaccines.48 However, as childhood respiratory infections are also a key risk factor for developing chronic respiratory diseases such as asthma and COPD,8 it is still important to improve vaccination rates overall across Europe through the collaborative efforts of healthcare providers and public health initiatives.
ANTIMICROBIALS IN THE PROPHYLAXIS AND TREATMENT OF RESPIRATORY INFECTIONS
Evidence for the benefit of antimicrobial agents for treating or preventing asthma exacerbations is limited. A recent RCT found no clinically significant benefit from azithromycin treatment in asthma attacks.49 Therefore, based on the limited data, prescribing antibiotics for preventing or treating asthmatic exacerbations is not recommended, unless there is strong evidence of lung infection.6
The use of antibiotics in preventing or managing COPD exacerbations is also controversial. Although COPD exacerbations are often treated with short-term (5–7 days) antibiotic use, there are conflicting data from clinical trials concerning the efficacy of this treatment. Indeed, a systematic review of 16 RCT in people with acute COPD exacerbations found antibiotics had no statistically significant benefit on mortality or hospitalisation duration.50
In terms of prophylactic use in stable disease, a 2013 Cochrane review of seven RCT concluded that the use of continuous prophylactic antibiotics (azithromycin, erythromycin, and clarithromycin) reduced the number of exacerbations in patients with COPD.51 Nonetheless, despite its benefits, it is estimated that >80% of COPD patients have risk factors for adverse complications from azithromycin therapy.52 Indeed, a study found those with chronic lung disease receiving long-term azithromycin therapy had an increased risk of hearing impairment and a 2.7-fold increased risk of colonisation with resistant bacteria relative to placebo controls, although azithromycin-treated patients had a lower risk of new colonisation (risk ratio: 0.55) and were less likely overall to be prescribed antibiotics for treatment (risk ratio: 0.63) compared with controls.53 In addition, a post-hoc analysis found patients with exacerbations requiring both antibiotic and steroid treatment, older patients, and those with milder disease (based on GOLD stage) may benefit from azithromycin treatment, while active smokers showed less benefit (i.e., no reduction in exacerbations of COPD was seen in this group).54 Therefore, antibiotic treatment regimens should be individualised and guided by the severity of symptoms, risk of exacerbations, and side effects.
Azithromycin (and potentially other antimicrobial agents) may be acting as immunomodulators. In smokers with emphysema, azithromycin therapy was shown to cause changes in the composition of the lower airway microbiota, which in turn increased the levels of bacterial anti-inflammatory metabolites, and subsequently decreased the levels of host-produced inflammatory chemokines (tumour necrosis factor-α, interleukin [IL]-12 p40, IL-13, and CXCL1).55 However, further investigation of the underlying mechanisms of the therapeutic effects of azithromycin in COPD is required.
Inhaled antibiotics have also recently gained attention for the prevention of COPD exacerbations. Patients with severe COPD colonised by multidrug-resistant Pseudomonas aeruginosa who were treated with nebulised tobramycin (twice daily for 14 days) showed reductions in local inflammation and bacterial load, with a concurrent 42% reduction in severe exacerbations after 6 months of treatment.56 The use of inhaled antibiotics for managing COPD exacerbations, however, requires further study.
Overall, at present, there is no Grade A evidence (RCT; rich body of data) on which GOLD or GINA base their recommendations regarding the role of antibiotics in preventing or treating infections during COPD and asthma exacerbations.5,6
The Importance of Antimicrobial Stewardship
Despite the aforementioned lack of evidence, it is estimated that >80% of COPD patients in secondary care and 50% of patients managed in primary care are treated with antibiotics.57 This is concerning as antibiotic use in patients with COPD may be contributing to antimicrobial resistance (AMR). To respond to the increasing threat of AMR, antimicrobial stewardship programmes have been developed in many countries worldwide (e.g., the UK One Health Report58).
Antimicrobial stewardship programmes aim to educate healthcare professionals to use an evidence-based approach when prescribing antibiotics, which will change according to the patient’s individual situation. Unfortunately, guidelines regarding the prescription of antibiotics in COPD and asthma are, at present, poorly followed, particularly in the case of recurrent exacerbations.59 For example, it is recommended that sputum diagnostics should be implemented in patients with early treatment failure or repeated exacerbation as soon as antibiotic treatment is started; however, at present, sputum cultures are only performed in a minority of cases.59 Moreover, expectorated sputum specimens are subject to significant contamination by oropharyngeal bacteria, making it difficult to distinguish between true pathogens and contaminants in cultured specimens. Newer diagnostic tests are being developed to describe the entire population of bacteria in bronchoalveolar lavage specimens, as there is mounting evidence that exacerbations are actually due to a pathological change in the composition of the population of microbes at the site of infection, rather than the presence of a single pathogen.57
To help reduce the impact of frequent antibiotic use in COPD on AMR, more research is required to identify which patients would benefit from antibiotics (such as the study by Han et al.54 on daily azithromycin use). Developing novel or more effective vaccines to prevent airway infections may also help reduce the emergence of multidrug-resistant strains. This was highlighted in a 2016 report commissioned by the UK Prime Minister,60 which concluded that we are not moving anywhere near fast enough to develop or use vaccines to reduce AMR. Preventing airborne infections in healthcare settings is another key strategy. The infection protection and control interventions available to prevent airborne infections include handwashing, healthcare worker vaccination, vaccination of patients, patient isolation, antiviral treatment, and the use of facemasks.61 Blanco et al.61 estimated that combining the above interventions in a bundle had the potential to reduce influenza transmission in a hospital by up to 50%.
CONCLUSIONS
As chronic airway diseases are largely incurable, prevention through reducing exposure to risk factors such as tobacco smoke is our best strategy. However, as summarised in Table 2, we also have a number of cost-effective interventions to reduce the symptomatic burden of these disorders to maintain the long-term respiratory health of patients with COPD or asthma. These strategies include smoking cessation; reducing exposure to allergens, pollutants, and biomass fuels; and infection control through vaccines and antibiotics. Regarding antibiotics, it is becoming increasingly necessary for all healthcare workers to make better choices regarding their use in respiratory infections, by relying on new diagnostics and adhering to antimicrobial stewardship programmes. As we enter a post-antibiotic era, the discovery of new and improved vaccines, and appropriate evidence-based vaccination schedules, is crucial for preventing not only the development of COPD and asthma but also the number of exacerbations and disease progression. By implementing the strategies reviewed here, we can support the vision of the World Health Organization (WHO) to create a world where all people can breathe freely.
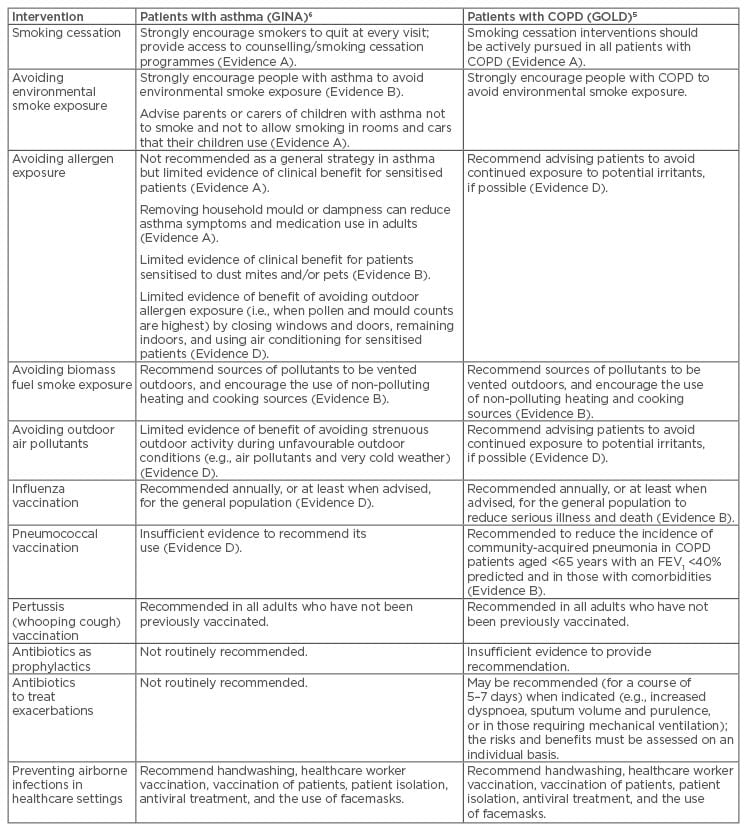
Table 2: Interventions other than bronchodilators and inhaled steroids to reduce the burden of asthma and chronic obstructive pulmonary disease.
Evidence A: randomised clinical trials (rich body of data); Evidence B: randomised clinical trials (limited body of data); Evidence C: non-randomised trials (observational studies); Evidence D: panel consensus judgement.
COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in one second; GINA: Global Initiative for Asthma; GOLD: Global Initiative for Chronic Obstructive Lung Disease.







