BACKGROUND AND AIMS
The diagnosis of pudendal neuropathy is based on a set of five criteria; four of them are clinical and the last one involves the evaluation of the response to CT-guided infiltration of the pudendal nerve.1 These criteria can be considered as cornerstones in the diagnosis of pudendal neuralgia (PN) and in the indication of decompression surgery (Nantes criteria). The objective of the present study was to analyse the efficacy of CT-guided pudendal nerve infiltration and its role in the PN diagnosis, and to identify predictive factors for response to CT-guided pudendal nerve block.
MATERIALS AND METHODS
A cross-sectional study was performed. Fifty-two subjects with PN who met the five essential Nantes criteria were identified. Thirty-seven patients with at least 1-year of follow up and who underwent CT-guided perineural injection were included. Pain response was assessed using a visual analogue scale (VAS). The remaining 15 patients were excluded because of one of the following two reasons: either a short follow-up period (<1 year) or the patient rejected undergoing diagnostic CT-guided injection. Injection responses were categorised as positive or negative, according to self-reported pain relief (VAS score) after the procedure.
Patients were evaluated for treatment outcomes (pain relief, duration, VAS change, and indication for surgical decompression). Mann-Whitney U, chi-squared, or Fisher’s exact tests, as well as logistic models were used to assess associations between covariates and positive response to CT-guided perineural infiltration or improvement after surgical decompression.
RESULTS
Overall, 45/52 (86.54%) patients were female and the mean age was 51.17 years (standard deviation [SD]: ±14.15 years). The response rate after CT-guided pudendal injection was 75.68% of patients. The mean duration of pain relief was 9.05 days (SD: ±11.59 days). The mean VAS scores were 7.66 (SD: ±1.50) and 3.00 (SD: ±2.44) before and after the procedure, respectively.
A diagnostic neurophysiological study was associated with the pain relief after CT-guided nerve block (p=0.02) (Table 1).
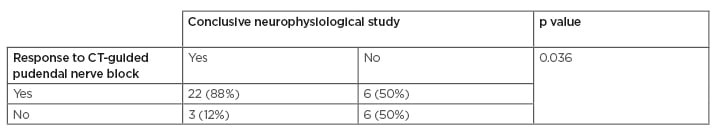
Table 1: Association between the efficacy of CT-guided pudendal nerve block and a conclusive neurophysiological study.
Multivariate analysis confirmed that a conclusive neurophysiologic testing was associated with the positive response to CT-guided pudendal nerve block (odds ratio: 9.54, range: 1.32–68.83; p=0.025).
Twenty-two patients (42.31%) underwent pudendal nerve decompression surgery. Of these, 15/22 (68.18%) patients improved after surgery. A favourable response to injection was associated with the surgical indication (p=0.002). In addition, 14/19 (73.68%) of patients who had a good response to CT-guided nerve block improved after surgical decompression, as well as those who had a positive prior neurophysiologic study (12/16; 75%).
CONCLUSION
CT-guided pudendal nerve block is an effective treatment option and plays an important role in the diagnosis of PN. The previous conclusive neurophysiological study is associated with the success of the CT-guided infiltration and can be used as a tool for predicting the response to this technique.








