Abstract
Premature ejaculation (PE) can be a very distressing condition and has been studied for many years. However, there exists confusion about the definition, incidence, and management of this condition. Treatment through pharmacotherapy has been focussed on topical agents along with oral antidepressant medications. The use of sexual psychology can also play a role in treatment of PE, often when added to medical therapies. Other alternative medical treatments have also been used with mixed outcomes. Although there is no perfect treatment for PE that works for every patient every time, there is ongoing research for the optimal therapy for men who complain about this problem. A review of the current understanding and medical management of PE will be set in this paper along with potential future treatments.
INTRODUCTION
Premature ejaculation (PE) was first described by Gross in 18871 and is thought to be one of the most common sexual dysfunctions affecting men today.2 However, the exact definition, epidemiology, pathophysiology, and management of PE have been disparate over time.3 The past several years have seen an increasing amount of research on PE, which has continued to change our perception regarding this condition, along with its management.3,4 Several professional organisations, such as the American Psychiatric Association (APA) and the International Society for Sexual Medicine (ISSM), have updated their definitions of PE to reflect the currently accepted and more evidence-based findings pertaining to PE.5,6 The newer and more concrete definitions allow for a better understanding and study of PE.
Today, a basic definition of PE is the following: i) an ejaculation that occurs in <1 minute from vaginal penetration (primary or lifelong PE) or <3 minutes (secondary or acquired PE); ii) ejaculation that cannot be postponed in nearly all attempts at vaginal penetration; and iii) personal distress with negative consequences such as avoidance of sexual relations.6 Despite our improved understanding of what truly constitutes PE, the optimal management of this condition remains a matter of debate. In the last century, the main treatment has consisted of various psychotherapy options.7,8 However, discoveries on the central regulation of ejaculation and the therapeutic role of serotonergic agents in PE revolutionised the treatment of this condition.9 This article aims to summarise the pathophysiology of PE and discuss its current medical treatment alternatives in the light of the most recent publications. We have conducted a search of the PE pharmacotherapy literature using the PubMed and Medline databases in January 2017, limited to original research publishedin English. Reference lists of identified articles were also reviewed for relevant publications.
PATHOPHYSIOLOGY OF PREMATURE EJACULATION
Ejaculation is thought to be controlled by many different physical pathways. The spinal ejaculatory generator (SEG),10 neuroendocrine issues,9,11 along with comorbid urological conditions12-14 can all affect ejaculation. The SEG works in concert with parasympathetic, sympathetic, and motor output in emission and expulsion phases of ejaculation.10,15 When certain thresholds of stimulation are met on the visceral, proprioceptive, and somatosensory neuronal input to the SEG and supraspinal sites, activation or inhibition of ejaculation occurs.15
Although we understand how ejaculation occurs, we still do not fully know the mechanisms that contribute to the pathology of PE. It has been theorised that hyposensitivity of the 5-HT2C or hypersensitivity of the 5-HT1A receptors could be the cause of lifelong PE.16 The same authors proposed that men with low serotonin may have a lower ejaculatory set-point resulting in PE. This theory would also help to explain delayed ejaculation in those with a genetically higher set-point.17 Hormones are known to play a role in ejaculation and timing, such as dopamine, oxytocin, testosterone, and prolactin.18,19 Additionally, comorbid urologic conditions like prostatitis can be associated with PE.12,20 Varicoceles have also been thought to contribute to pelvic venous congestion and symptomatic prostatitis resulting in PE,21 wherein after surgical correction of the varicocele, symptoms of PE were improved.13 Certainly the myriad of biological causes of PE are not well elucidated, but we are gaining a better understanding of the condition which may help guide future treatment options.
The contribution of the psychological factors to the biological ones makes the pathophysiology of PE even more complex. Psychologically, men with PE have high rates of dissatisfaction with sexual intimacy and relationships.22-24 The partners of men with PE were studied using a validated sexual questionnaire, which reported that the female partners were also negatively affected with decreased sexual satisfaction when they had sex with a man who has PE.25,26 One theory suggested that men with PE had increased sympathetic activities due to anxiety which promotes smooth-muscle contraction resulting in PE.27 There is some psychopathology associated with PE in the form of decreased self-esteem and self-confidence, increased interpersonal problems, mental obsession with ejaculation and performance, and increased embarrassment/shame/guilt/worry about sexual failure and the inability to please the partner.23,24 Therapies directed at treatment of these comorbid conditions, which may be the result or the cause of PE, are important prescriptions in the healing from PE.
PSYCHOTHERAPY FOR PREMATURE EJACULATION
The first treatment modalities for PE were with psychotherapy focussing on the sexual relationship and anxiety related to sexual satisfaction with a partner.28,29 A behaviour modification technique was popularised by Master and Johnson30 as they described the ‘squeeze’ and later ‘start-stop’ method. This is a form of sensory behavioural therapy, which is thought to train the mind and body around ejaculatory timing, psychological excitation, and stimulation responses.31 However, these types of techniques have not reliably been able to increase the intravaginal ejaculatory latency time (IELT).28,31,32
Psychotherapies directed at increasing sexual knowledge, relationship strengthening, interpersonal resolution of sexually related disorders, treatment of psychopathologies that may lead or result in PE, increasing sexual skills set, and reduction of performance anxiety, have been suggested as helpful treatment regimens for PE.33,34 However, many psychological studies are not strongly evidence-based and have methodological flaws and are hence considered to be adjuvant treatments along with pharmacotherapy.33,35 In two meta-analyses published more recently, it was shown that the intervention with psychotherapy was inconsistent, which means more research is needed to assess efficacy.36,37 Until more quality research is produced, it is advised to use psychotherapy in combination with pharmacotherapy for treatment of PE.
PHARMACOTHERAPY FOR PREMATURE EJACULATION
Topical Anaesthetics
In 1943, the first medical treatment for PE was suggested by Schapiro.29 He described how topical anaesthetics may help to delay ejaculation by decreasing the hypersensitivity of the glans penis during sexual intercourse as hypersensitivity of the glans is one of the proposed aetiological factors in the pathophysiology of PE.38
The basic topical medications used to treat PE tend to be lidocaine based.39 A trial of 5% lidocaine-prilocaine cream increased the IELT when it was applied 20 minutes prior to sex in a randomised, placebo-controlled double-blind trial.40 A study that looked at a combination of lidocaine-prilocaine cream versus a phosphodiesterase Type 5 inhibitor (PDE5-I) showed that the topical agent worked much better in patient satisfaction rates and IELT times for PE.41 Administration changes of topical agents have showed success with IELT in PE. There is a topical spray mixture for PE known as TEMPE Spray (Plethora Solutions Ltd, London, UK) that contains lidocaine-prilocaine which increased IELT by 6.3-times compared to baseline with excellent patient satisfaction and reported outcomes.42 There have also been more natural products that have had good outcomes in terms of success in topical application for treatment of PE. One of these is known as Severance-Secret (SS)-cream and is a mixture of nine herbal supplements.39 One study of SS-cream demonstrated an increase of baseline IELT from 1.37 to 10.92 minutes with reported sexual satisfaction rates of 82% in a controlled study.43 However, with every treatment comes some adverse effects. With the topical anaesthetics, there have been reports of hypoaesthesia in the partner, thought to be from direct transfer of the topical agent to the vagina. If a condom is not used, this transfer can result in vaginal anaesthesia and anorgasmia in the female.44
Selective Serotonin Reuptake Inhibitors
When the regulation of serotonin is disrupted in the central nervous system, ejaculation can be altered, resulting in one of the proposed aetiologies of PE.45,46 From studies in animals and humans, it has been determined that the serotonin neurotransmitter has the greatest impact on ejaculation and timing.16,47 Because of these findings, selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants and selective norepinephrine, and serotonin reuptake inhibitors have all been used in the management of PE.48 These drugs prevent the reabsorption of serotonin from the axonal end within the synaptic cleft. This results in an increase in serotonin within the microneuronal environment and increased serotonin stimulation in the post-synaptic receptors. Increasing serotonin levels then results in a delay of ejaculation. Dapoxetine is a fast-acting SSRI that can be used to treat PE on demand and it is often considered as the first-line drug therapy.49 With the recently developed delivery methods, dapoxetine appears to have an even quicker onset of action and bioavailability from sublingual and intranasal routes.50 Dapoxetine is not approved for use in the USA and paroxetine has been used on an on-demand basis or daily as it has a relatively greater efficacy in delaying ejaculation.51,52 Other SSRIs would need to be used daily (paroxetine, clomipramine, sertraline, fluoxetine, or citalopram) (Table 1).46,53-60
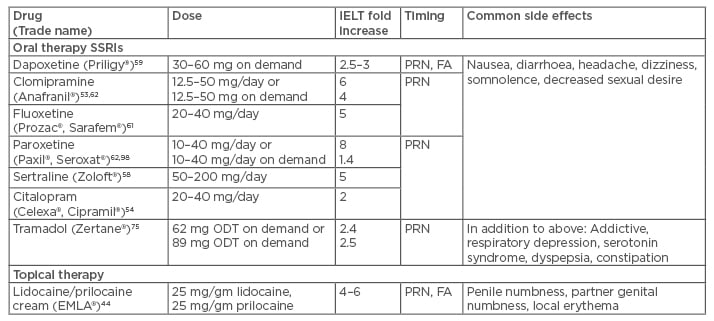
Table 1: Medical treatment options for premature ejaculation.97
SSRI: selective serotonin reuptake inhibitors; IELT: intravaginal ejaculatory latency time; PRN: as needed; FA: fast acting (quick on-set of action); ODT: orally dissolving tablet.
The doses of SSRIs that are effective in increasing IELT are paroxetine 10–40 mg, clomipramine 12.5–50 mg, sertraline 50–200 mg, fluoxetine 20–40 mg, and citalopram 20–40 mg (Table 1).53-55,58,60,61 It has been shown that paroxetine has been the most effective with an 8.8-fold increase in IELT compared to the other SSRIs.62 Generally, it takes time for these drugs to assert their effect. However, some may seem to affect ejaculation within 5–19 days, with the maximal effect usually being reached within 2–3 weeks of daily treatment.63 The side effects of SSRI treatments are relatively minor and may even dissipate after 2–3 weeks of continued use.64 The most common side effects are mild nausea, diarrhoea, yawning, constipation, insomnia, and fatigue.64 Some degree of sexual dysfunction in the form of erectile dysfunction, low libido, or hypoactive desire was reported with SSRI use for PE.65 Additionally, patients who are interested in fertility should use SSRIs with caution as there has been evidence of decreased sperm parameters with SSRI use.66
Perhaps the most serious side effect of SSRI use is the suicide risk which may occur in the early phase of the treatment for young adults, adolescents, and children with concurrent psychiatric disorders.67 Suicidal thoughts should be appropriately screened with a low-threshold for referral to psychiatric care if needed. In adult men with PE, such risk was not very significant, but clinicians should still exercise caution when prescribing such medications.51,59
When evaluating the compliance of SSRIs for patients being treated for PE, there seems to be a 30% incidence of non-starters for paroxetine (10 mg daily for 21 days followed by 20 mg as needed) and another 30% who did not follow-through with treatment after they had started, citing that the side effects of treatments seemed to be worse than the PE condition (treatment less effective than expected, fear of being on an antidepressant, loss of interest in sex from relationship, or drug side effects).68 There have not been a lot of side effects from the use of dapoxetine on demand dose, but those that were reported included headaches, dizziness, and nausea, and were dose-dependent.59,69 Although the majority of lifelong PE patients prefer daily drug treatment,70 some men may be willing to use on-demand dapoxetine because of the potential side effects of continuous SSRI use. However, it has been shown that about 20% of men do not want to be on any drug due to fear of side effects or cost and hence never start treatment with dapoxetine.71 In fact, 90% discontinued dapoxetine within a year for a number of reported reasons: expected outcome less than hoped for (24.4%), price of the drug (24.4%), decreased libido or sex drive felt to be from the drug (19.8%), and the drug not being effective to treat the condition (13.9%).71
Tramadol
Another drug that has been used to help with PE is tramadol. This drug can be used as needed or daily to help with PE by increasing the IELT.72 The mechanism of action of tramadol is to bind the μ-opioid receptors and weakly inhibit the serotonin/ norepinephrine reuptake receptors, which results in analgesia.73 Tramadol in doses of 25–100 mg has been shown to increase the IELT by 2.4 to 12.6-fold in controlled clinical trials.74,75 Unfortunately, as effective as tramadol can be, it can also have severe negative consequences including drug addiction, somnolence, pruritus, dizziness, dry mouth, vomiting, and nausea, which can all be dose-dependent.74,75 Additionally, the combination of tramadol with an SSRI can be fatal due to induction of a serotonin syndrome and should therefore be avoided.76
Phosphodiesterase Type 5 Inhibitors
PDE5-Is have been well-studied and may marginally help those who have comorbid erectile dysfunction and PE.77 Some men have conditioned themselves to have a more rapid ejaculation due to the inability to maintain erections and for this population it is proposed that PDE5-Is may be helpful to prolong the sexual experience, but there is no robust evidence that have supported the use of PDE5-Is in PE patients who do not have concomitant erectile dysfunction.
Other Drugs and Treatments
Oxytocin and dopamine are two neurochemicals that effect ejaculation and which have been used for some therapeutic benefit in ejaculatory dysfunction.11,78 Oxytocin has been shown to shorten ejaculatory latency periods and decrease the refractory period as well in both animals and humans.79.80 Studies have also been done on rats, which demonstrated that an oxytocin antagonist resulted in delayed ejaculation.81,82 One promising trial used an oxytocin antagonist to help prolong ejaculation while acting on peripheral and central oxytocin receptors.78 Further clinical studies are needed to support oxytocin manipulation treatments for PE.
Alpha-1 adrenoreceptors seem to play role in the ejaculation process83 and inhibiting their function with alpha blockers (silodosin, tamsulosin hydrochloride, alfuzosin, terazosin, and doxazosin) is hypothesised to be an effective treatment for PE.84 Among these drugs, silodosin is associated with higher risk of abnormal ejaculation,85-87 as 14% of subjects treated with silodosin reported a reduction or absence of ejaculation compared to tamsulosin (2.1%) and placebo (1.1%).88 Sato et al.89 evaluated the feasibility of off-label silodosin (4 mg) treatment in eight patients with PE and demonstrated that this drug has an ejaculation-delaying effect (from 3.4 to 10.1 minutes, p=0.003) when it is taken 2 hours before the planned sexual intercourse. These results support the possible use of α1-adrenoceptor antagonists as a new treatment option for PE; however, further placebo-controlled trials assessing this application are needed.
Botulinum neurotoxin, which is produced by Clostridium botulinum, is a highly potent biomolecule which prevents the release of the acetylcholine from the neuronal endings at the neuromuscular junction and causes flaccid paralysis.90 It has been hypothesised that paralysing the muscles which are involved in the ejaculation process (i.e. bulbospongiosus and ischiocavernosus muscles) may delay ejaculation latency, thus ameliorating the PE condition.91 It was also recently demonstrated that botulinum-toxin injection into the bulbospongiosus muscle in rats was associated with an increase in the ejaculatory latency without affecting the ability to achieve erection or ejaculation.92 However, clinical studies to demonstrate the efficacy and safety of this treatment modality in men with PE are warranted.
Modafinil is an oral wakefulness-promoting drug that is mainly used for the treatment of narcolepsy.93 It exerts this effect by interacting with norepinephrine, serotonin, dopamine, and GABA-containing neuronal systems. In addition to its stimulant effect on wakefulness, it has been demonstrated that it can increase the ejaculation latency in rats without suppressing sexual behaviour.94 This effect has been confirmed in a case study of a lifelong PE patient,95 whereas Tuken et al.96 demonstrated that on-demand modafinil treatment significantly increased mean IELT and improved patient reported outcomes in 55 lifelong PE patients. Future controlled clinical trials are necessary to confirm these findings.
CONCLUSION
Pharmacotherapy for PE tends to be 2-fold: topical application of anesthetic agents and daily or on-demand SSRIs. All pharmacologic agents have some side effects, which can affect patient compliance to treatment. Tramadol and PDE5-Is have some therapeutic effect but much less than the proven therapies. C. botulinum toxin and modafinil are promising pharmacologic agents for PE treatment, but their efficacy must be confirmed in well-designed clinical trials. The future of PE pharmacotherapy is beginning to flourish with research. Ideas on drug delivery, understanding of ejaculatory physiology and hormones, as well as uses of various pharmacologic agents are paving the way for promising future PE treatments.








