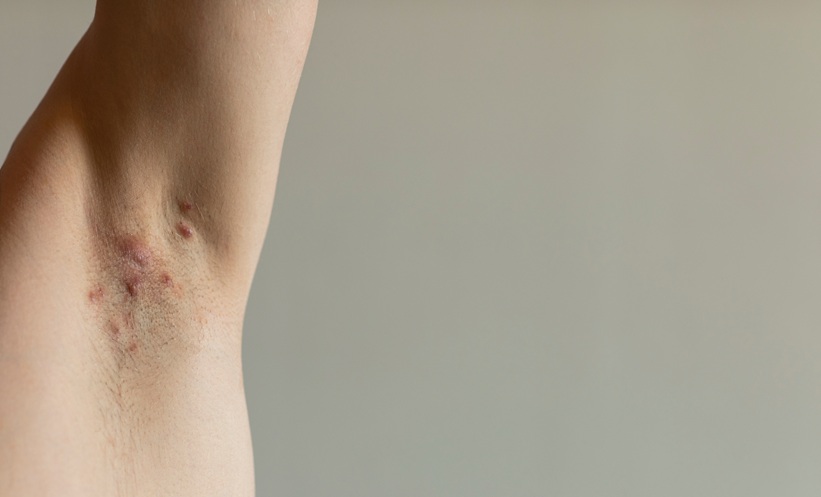
EMERGING evidence suggests hidradenitis suppurativa treatment may soon expand, as a phase 2 trial demonstrates that lutikizumab improves clinical outcomes in patients who previously failed anti-TNF therapy, offering a potential new targeted option for this difficult to treat condition.
Unmet Need in Hidradenitis Suppurativa Treatment
Hidradenitis suppurativa treatment remains challenging, particularly in patients with moderate to severe disease who do not respond to anti-tumour necrosis factor therapies. The chronic inflammatory condition is associated with significant pain, recurrent lesions, and impaired quality of life, yet therapeutic options remain limited. Interest has therefore grown in targeting interleukin pathways, particularly IL-1α and IL-1β, which are implicated in disease pathogenesis. Lutikizumab, a dual-variable domain antibody, has been developed to inhibit both cytokines simultaneously, raising the possibility of a more effective hidradenitis suppurativa treatment for refractory populations.
Trial Design and Key Results in Hidradenitis Suppurativa Treatment
In this phase 2, double blind, placebo-controlled trial, 153 participants received at least 1 dose of study medication (94 female individuals [61.4%]; mean [range] age, 40.5 [19-75] years; 108 patients [70.6%] at Hurley stage III). At week 16, 19 (48.7%), 22 (59.5%), and 10 participants (27.0%) receiving lutikizumab, 300 mg, every week; lutikizumab, 300 mg, every other week; and lutikizumab, 100 mg, every other week, respectively, achieved HiSCR50 at week 16 compared with placebo (35.0%). Among participants with baseline NRS scores of 3 or greater, 10 (34.5%) and 8 (34.8%) in the higher dose groups achieved NRS30 response compared with 4 (12.9%) receiving placebo. Serious adverse events occurred in 2 of 39 participants (5.1%) receiving weekly dosing, 2 of 37 (5.4%) receiving biweekly dosing, and 1 of 40 (2.5%) receiving placebo.
Implications for Future Hidradenitis Suppurativa Treatment
These findings suggest that dual IL-1 inhibition could represent a meaningful advancement in hidradenitis suppurativa treatment, particularly for patients with limited alternatives after biologic failure. Although the study was not powered for definitive conclusions, consistent improvements across endpoints and a manageable safety profile support further clinical development. Future larger trials will be essential to confirm efficacy, optimise dosing strategies, and determine long term safety. If validated, lutikizumab could contribute to a more personalised approach to hidradenitis suppurativa treatment, helping clinicians better address disease heterogeneity and improve patient outcomes.
Reference
Kimball AB et al. Lutikizumab in adults with moderate to severe hidradenitis suppurativa after Anti-TNF therapy failure: a phase 2 randomized clinical trial. JAMA Dermatol. 2026;DOI:10.1001/jamadermatol.2026.0155
Featured image: Lea on Adobe Stock