BACKGROUND AND AIMS
Acute myocardial infarction (AMI) complicated by cardiogenic shock is associated with significant morbidity and mortality. Mechanical circulatory supporting devices, including microaxial left ventricular assist devices (MLVAD; Impella®, Abiomed, Danvers, Massachusetts, USA, and TandemHeart®, LivaNova, London, UK) and intra-aortic balloon pumps (IABP), have increased treatment options in addition to pharmacotherapy. IABP consist of a cylindrical balloon placed in the aorta and work via ‘counterpulsation’ with balloon inflation in diastole and deflation in early systole; however, the IABP-SHOCK II trial showed inferior survival and more complications compared with optimal medical treatment.1 On the other hand, MLVAD are positioned across the aortic valve via a peripheral artery and create continuous forward blood flow from the left ventricle into the aorta at a constant rate by a rotary pump. Despite that, all randomised clinical trials, including PROTECT II, ISAR-SHOCK, IMPELLA-STIC, and IMPRESS in Severe Shock, were not able to discern survival benefit over IABP.2,3 There is limited knowledge of the in-patient complications of these devices.
Purpose
To compare the outcomes of patients with AMI who underwent percutaneous coronary intervention (PCI) complicated by cardiogenic shock treated with IABP versus MLVAD.
MATERIALS AND METHODS
The Nationwide Inpatient Sample (NIS) database is the largest inpatient registry in the USA. The authors used NIS data from 2009–2014 to identify adult patients admitted for AMI who received PCI complicated by cardiogenic shock. Based on the use of IABP or MLVAD, the study population was divided into two groups. To reduce selection bias, propensity score matching was performed using the Kernel method. Patient characteristics, hospital characteristics, and comorbidities were matched. Logistic regression was used for categorical variables including in-hospital mortality, requirement for blood transfusion, sepsis, cardiac arrest, and cardiac complications (including iatrogenic complications, haemopericardium, and cardiac tamponade). Poisson regression was used for continuous variables, including length of stay and total cost.
RESULTS
A total of 49,837 patients were identified. With propensity score matching, 34,132 patients in the IABP group were matched to 1,430 patients in the MLVAD group. Compared with the MLVAD group, the IABP group had lower in-hospital mortality rates (28.29% versus 40.36%; odds ratio [OR]: 0.58 [0.42–0.81]; p=0.002), lower rates of blood transfusion (9.63% versus 11.50%; OR: 0.49 [0.27–0.88]; p=0.017), and lower costs ($47,167 versus $70,429 USD; p<0.001). The IABP and MLVAD groups had similar lengths of stay (8.9 versus 9.3 days; p=0.882), rates of cardiac complications (6.50% versus 7.24%; OR: 0.56 [0.26–1.19]; p=0.134), rates of sepsis (9.30% versus 14.98%; OR: 0.66 [0.38–1.14]; p=0.133), and rates of cardiac arrest (37.84% versus 41.05%; OR: 0.70 [0.45–1.10]; p=0.123). (Table 1)
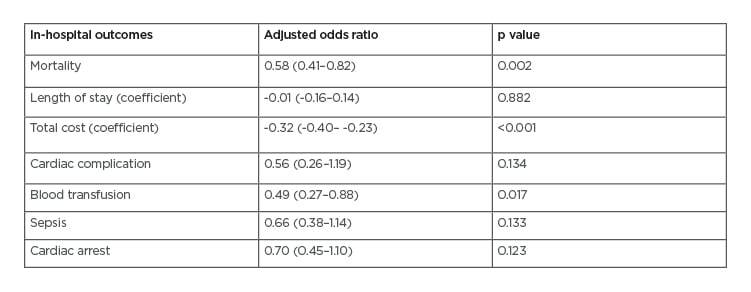
Table 1: Propensity score-matching outcomes.
CONCLUSION
In patients with AMI who underwent PCI complicated by cardiogenic shock, MLVAD compared with IABP was associated with higher risk of in-hospital mortality; greater requirement for blood transfusion, indicating the presence of major bleeding complications; and higher cost, although study interpretation is limited by the retrospective observational design. Further research is warranted to elucidate the optimal mechanical circulatory supporting devices in these patients.








