BACKGROUND
Several studies have demonstrated the benefits of transcatheter aortic valve implantation (TAVI) in high and intermediate-risk patients, but there is still a gap in the evidence regarding the long-term outcomes related to pharmacological therapies.1-3 In particular, the presence of fibrosis and myocardial hypertrophy in patients with aortic stenosis has been related to worse prognosis.4,5 Therefore, better outcomes might be achieved with the use of strategies that improve cardiac remodelling by reversing fibrosis and hypertrophy.6,7 In this regard, renin-angiotensin system (RAS) blockade has been shown to have a positive impact on remodelling and major clinical outcomes in previous studies.8-10 In this study, we aimed to determine the effects of RAS blockade following successful TAVI procedures.
METHODS
Patients with severe aortic stenosis who underwent TAVI between August 2007 and August 2017 were included from nine institutions. All baseline clinical, echocardiographic, and procedural data were prospectively recorded in a dedicated database and prespecified follow-up was performed. Dose and type of RAS blockade therapy were also recorded. Patients were compared according to the prescription of RAS blockade (or lack of) at discharge. Only those patients who survived the in-hospital period and who had been on continuous RAS blockade therapy for at least 1 month after TAVI were included in the treatment group. A matched comparison was performed according to baseline and procedural differences.
RESULTS
A total of 2,866 patients were included and the final study population consisted of 2,715 patients who were alive at discharge. The mean age of the study population was 80.8±7.1 years and 53.7% of the patients were female. A total of 1,622 patients (59.7%) received RAS blockade therapy after the procedure and at a median follow-up of 3 years post-TAVI, the RAS blockade group had significantly lower cumulative mortality than the non-RAS blockade group (9.5% versus 16.5%; p=0.001, log rank test). Matched adjustment by main baseline and procedural differences, including hypertension, diabetes, chronic kidney disease, chronic pulmonary disease, baseline New York Heart Association (NYHA) class, and baseline left ventricular ejection fraction, was performed. No differences existed between the use of other medications, the degree of residual aortic regurgitation, or the TAVI approach. RAS blockade therapy was still associated with significantly lower cardiac mortality (hazard ratio: 0.581; 95% confidence interval: 0.310– 0.893; p=0.002) both at 1 and 3-year follow-up (Figure 1). In addition, the rates of myocardial infarction, cerebrovascular events, and hospital readmission due to heart failure remained lower in the RAS blockade group than the non-RAS blockade patients.
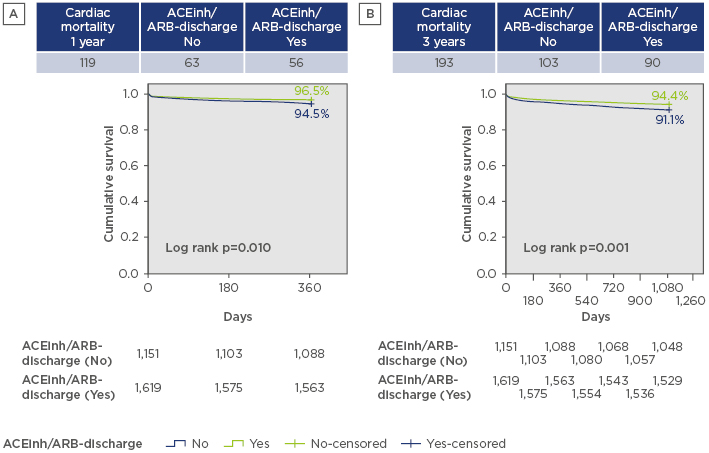
Figure 1: Cardiac mortality at A) 1 and B) 3-year follow-up following transcatheter aortic valve implantation with and without renin-angiotensin system blockade.
ACEinh: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker.
DISCUSSION
While a positive effect of RAS blockade was expected, the marked cardiovascular protective effect irrespective of other conditions is a major finding from this study. Although some authors suggest that many post-TAVI patients are already prescribed cardioprotective treatments, we found that up to 40% of patients were not. Confirmation of such effects and analysis of the cardioprotective mechanisms of RAS blockade are warranted and will be conducted through the RASTAVI clinical trial,11 which will investigate the use of ramipril for RAS blockade after TAVI for severe aortic stenosis.
CONCLUSION
This retrospective cohort of the randomised RASTAVI study suggests that post-TAVI RAS blockade therapy is associated with lower cardiac mortality at 3-year follow-up and results in a cardiovascular protective effect irrespective of the left ventricular ejection fraction or residual aortic regurgitation. The RASTAVI randomised trial will help to determine the accuracy of these findings.








