Interviewees: Renato D. Lopes,1 Amit N. Vora1,2
1. Duke Clinical Research Institute, Durham, North Carolina, USA
2. UPMC Pinnacle Heat and Vascular Institute, Harrisburg, Pennsylvania, USA
Disclosure: Prof Lopes has been a consultant to Bayer AG, Boehringer Ingelheim, Daiichi-Sanyo, Merck, and Portola; and has been a consultant and received grants from Bristol-Myers Squibb, GlaxoSmithKline, Medtronic, Pfizer, and Sanofi. Dr Vora has declared no conflicts of interest.
Support: Supported by an Educational Grant from Bristol Myers Squibb & Pfizer.
Acknowledgements: Medical writing assistance was provided by Dr Eleanor Roberts, Beeline Science Communications, Ltd, London, UK.
Disclaimer: The opinions expressed in this article belong solely to the named interviewees.
Interview Summary
For someone with nonvalvular atrial fibrillation (AF), co-occurrence of acute coronary syndrome (ACS), or need for percutaneous coronary intervention (PCI) can bring treatment dilemmas due to the need for both oral anticoagulation (OAC) therapy for AF and antiplatelet therapy for ACS/PCI. AUGUSTUS was the largest trial to date to investigate treatment of AF in those with ACS/PCI and was run with a unique 2×2 factorial design whereby participants were separately randomised to either the direct OAC medication apixaban or a vitamin K antagonist (VKA) and either aspirin or an aspirin placebo, all with a P2Y12 inhibitor. The inclusion in the trial of those who were ‘medically managed,’ without PCI was another unique factor that helped tease out how real-world patients with AF may be treated if they have ACS.
Prof Renato Lopes and Dr Amit Vora, clinical researchers involved in AUGUSTUS, discuss here how findings from the trial are changing the landscape of prescribing for people with AF and ACS or/and PCI. The main results of AUGUSTUS showed that the apixaban-based regimen was superior to a VKA-based strategy in terms of fewer hospitalisations and that, for many, as shown in earlier studies, addition of aspirin was unnecessary and potentially harmful, as evidenced by a lower number of bleeding incidents in the placebo aspirin group. For those undergoing PCI, AUGUSTUS showed that aspirin may be useful in the first 30 days only.
The findings of AUGUSTUS, in combination with previous studies, are set to change guidelines and practice regarding the best treatment regimen for someone with AF with ACS and/or PCI.
INTRODUCTION
AF can increase stroke risk and is generally treated with OAC therapy, such as with a VKA or a direct OAC (DOAC). However, what if someone with AF also experiences ACS and/or needs PCI? Management of ACS and/or PCI typically requires antiplatelet therapy, such as with aspirin and a P2Y12 inhibitor (P2Y12i). Balancing therapy requirements can be difficult and has historically been little investigated.
AUGUSTUS is, to date, the largest trial of treatment of nonvalvular AF with ACS and/or PCI. In a 2×2 factorial design, participants received either the DOAC apixaban (n=2,306) or a VKA (n=2,308), and either aspirin (n=2,307) or a placebo (n=2,307), on background P2Y12i therapy.1 AUGUSTUS was the only major trial of its type to include ‘medically managed’ ACS patients, who did not receive a stent, as well as those undergoing PCI. Prof Renato Lopes and Dr Amit Vora, clinical researchers involved in the trial, spoke with EMJ about how findings from AUGUSTUS are changing the landscape of prescribing for people with AF and ACS or/and PCI.
HOW HAVE GUIDELINES TRADITIONALLY ADDRESSED TREATMENT FOR THESE PATIENTS?
As there are separate guidelines for those with AF and those with ACS, “this is a difficult population to treat,” explained Dr Vora. “The cornerstone of treatment for AF is to reduce risk of stroke with OAC therapy, but this is not sufficient to prevent recurrent ischaemic events in those with ACS or PCI. Conversely, for ACS, guidelines recommend dual antiplatelet therapy, but this isn’t as good as an OAC for reducing overall stroke risk.” Dr Vora discussed how triple antithrombotic therapy (VKA, P2Y12i, and aspirin) has been the mainstay therapy for over 20 years, “despite data suggesting that bleeding risk is very high in these patients.” However, he said: “There was no good, meaningful way to study trade-offs. People did what they thought was best but without
clear guidelines.”
Prof Lopes explained that there are potentially 2.8 million treatment options for people with AF and ACS and/or PCI. Choice was hard because people with AF plus ACS “have been mainly excluded from trials as they are very high risk, they bleed a lot, and have a lot of ischaemic events.” Because of this, “guidelines have been largely driven by observational studies, consensus documents, and professional opinions [where] the level of evidence is not high with a low level of certainty of suggestions.”
“DOAC for AF are already a Class I recommendation and are better than VKA,” continued Prof Lopes, “but what we didn’t know is whether this is true for those who undergo PCI and have ACS, and who also require antiplatelet drugs. The big challenge is finding the right combination, at the right dose, for the right duration to reduce ischaemic events as much as possible while minimising bleeding risk. This is the antithrombotic sweet spot.”
FOUR TRAILS HAVE EXAMINED DIRECT ORAL ANTICOAGULATION USE IN THESE PATIENTS, WHAT ARE THE SIMILARITIES AND DIFFERENCES BETWEEN THEM?
The 2013 WOEST study2 showed significant efficacy and safety benefits with OAC therapy plus a P2Y12i compared to this regimen plus aspirin. “This was the first randomised study where people started to think about the potential for dropping aspirin in these patients,” said Dr Vora.
Four large trials1,3-5 (Table 1) have examined DOAC regimens for AF plus ACS. However, differences among the trials include not only DOAC type, but also dosing, therapy combination, and follow-up time, making them difficult to compare. One important difference is that in all but AUGUSTUS, participants needed to undergo PCI, meaning those medically managed with ACS were excluded. Prof Lopes explained that these patients are an important group as “not everyone with ACS has a PCI.” Dr Vora highlighted that in the PIONEER3 or RE-DUAL4 trials, there was lower bleeding in the DOAC arms but “you don’t know if that was because of the DOAC or because they didn’t use aspirin. The biggest strength of AUGUSTUS is the 2×2 factorial design.
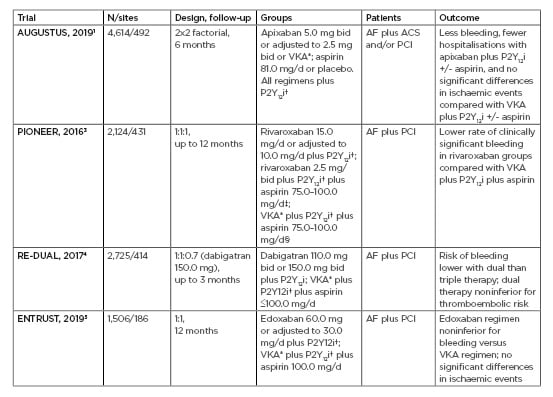
Table 1: Key aspects of the main clinical trials examining the use of direct oral anticoagulation.
*VKA dose adjusted to reach a target international normalised ratio of 2.0–3.0.
†Choice of P2Y12i was at the prescriber/study site discretion with dose according to drug.
‡After 1/6 months, approximately 100/250 in each group were switched to rivaroxaban 10 or 15 mg/day plus aspirin 75–100 mg/day.
After 1/6 months, approximately 100/250 in each group were switched to VKA* plus aspirin 75–100 mg/day.
+/-: with/without; ACS: acute coronary syndrome; AF: atrial fibrillation; bid: twice daily; mg/d: mg per day; N/sites: number of participants/number of study sites; PCI: percutaneous coronary intervention; P2Y12i: P2Y12 inhibitor; VKA: vitamin K antagonist.
These two independent arms really allowed us to study independent effects of aspirin, aspirin placebo, apixaban, and VKA.” Both clinicians pointed out that in PIONEER, rivaroxaban was used at a lower dose than the usual 20 mg/day, which, according to Prof Lopes, “may not be the most appropriate to use for this population. The key,” he continued, “is that we should use a dose shown to be effective for stroke prevention in AF.”
WHAT CLINICAL QUESTIONS DID AUGUSTUS ADDRESS AND HOW DID IT ADD TO CURRENT KNOWLEDGE?
Dr Vora described how a key finding of AUGUSTUS “is that it demonstrates that an apixaban-based strategy is superior to a warfarin or VKA-based strategy.” Additionally, Prof Lopes discussed how AUGUSTUS was the only trial that answered how many fewer bleeding incidents there were “by avoiding aspirin independently of DOAC use.” He explained that treating patients with a DOAC, at the right dose, with a P2Y12i, and without aspirin “gives us the closest regimen to the sweet spot combination of drugs where there is less bleeding and fewer hospitalisations, without any difference in ischaemic events.”
WHAT ARE THE KEY LEARNINGS ABOUT STENT THROMBOSIS FROM THIS TRIAL?
A subanalysis of AUGUSTUS examined stent thrombosis (n=30) in those undergoing PCI (n=3,498).6 This showed that there were nominally fewer stent thrombosis incidents in the apixaban arm (n=13) compared to VKA (n=17), but without significant differences. There were numerically higher counts of stent thrombosis in nonaspirin-treated patients (n=19) compared to aspirin groups (n=11), which, Dr Vora highlighted, is important because it shows “there may be an ischaemic benefit to continuing aspirin, although this needs to be balanced against the significant bleeding risk this entails.”
Prof Lopes discussed how “when you look at the trade-off between major bleeding and stent thrombosis, in the first 30 days you have a relationship that’s close to 1:1.” This means that for those with a higher stent thrombosis risk and low risk of bleeding, “it’s reasonable to consider keeping aspirin for 30 days in addition to apixaban and clopidogrel, then after 30 days you should stop aspirin as it doesn’t significantly prevent any more ischaemic events but increases bleeding.”
“The first 30 days are key,” said Dr Vora, “because if bad things are going to happen, they generally happen then.” This occurred, he explained, especially in those who had received a long stent or if it was put into a lower calibre vessel and where stent thrombosis location would be particularly catastrophic (e.g., in the left main coronary artery). For these patients, Dr Vora said he “would think about being more robust in terms of therapy, this includes aspirin, even if it modestly increases bleeding risk.”
WHAT WERE THE FINDINGS REGARDING HOSPITALISATIONS IN THE AUGUSTUS TRIAL?
Another AUGUSTUS subgroup analysis investigated hospitalisations.7 Prof Lopes discussed how they learnt that overall, the main causes of hospitalisation were cardiovascular causes, not bleeding. He highlighted how apixaban reduced hospitalisation for cardiovascular, bleeding, and all-cause-related hospitalisations compared to VKA and how using aspirin increased only bleeding-related hospitalisations.
“Practically,” said Dr Vora, “hospitalisations tend to be expensive. Warfarin is cheap, DOAC tend to be more costly, so we were looking at other significant benefits such as increased safety and efficacy, and the need for international normalised ratio monitoring. If DOAC can significantly reduce hospitalisation risk, the overall cost/benefit may be good.”
WHAT ABOUT MEDICALLY MANAGED VERSUS PERCUTANEOUS CORONARY INTERVENTION PATIENTS IN AUGUSTUS?
AUGUSTUS was important because it was the only trial to include people with ACS who did not undergo PCI. “The formal recommendation for medically managed ACS is 12 months dual antiplatelet therapy,” explained Dr Vora. This means that there is the same therapy-balancing conundrum as for those who receive a stent. Medically managed patients benefit from dual antiplatelet therapy but are “probably not being treated in quite the same way because providers may think ‘they don’t have a stent, so they only need aspirin’ when probably that isn’t the
right strategy.”
One subanalysis of AUGUSTUS investigated differences between those with AF with medically managed ACS (n=1,097), ACS and a PCI (n=1,714), or an elective PCI (n=1,784).8 Prof Lopes highlighted how the main analysis results were preserved regardless of medically managed or PCI treatment. “This was important as it closed the loop about the safety and efficacy of apixaban in AF patients across the spectrum of coronary artery diseases. It gives us extra confidence that we can apply the main AUGUSTUS results to this patient population. You can’t say anything about this population based on the other trials.”
WHAT DO YOU THINK THE IMPACT OF THIS TRAIL WILL BE ON FUTURE GUIDELINE UPDATES AND CLINICAL PRACTICE?
“Until 4 years ago,” underlined Prof Lopes, “guidelines were based on expert opinions and low-quality studies. Now, we have four randomised trials that give us close to 12,000 patients’ worth of data and we have consistent results and a message: less is more, select a NOAC [novel OAC] at the right dose, use a P2Y12i, stop aspirin at hospital discharge (or after the first 30 days for some patients), and avoid triple therapy with warfarin unless it’s the only option because otherwise you only gain harm without any additional benefits.” Dr Vora added that “this study will be practice-changing, the other studies laid the groundwork for at least thinking about no more aspirin in these patients, AUGUSTUS really confirmed that message.”
CONCLUDING REMARKS
According to Prof Lopes: “The key point of AUGUSTUS is that it examined a field of patients that exist quite commonly in clinical practice. Although we don’t have all the answers yet, and we haven’t looked at all 2.8 million antithrombotic treatment possibilities, we have studied a few of them and got some of the directions based on high-quality evidence that should be applied in clinical practice.” Dr Vora concluded that “AUGUSTUS is a landmark study that allowed us to determine the optimal strategy for these patients. It is very important in that it clearly answered the question [of how to treat these patients] in a definitive manner, which is all you can hope for from any clinical trial.”








