Abstract
Takotsubo syndrome is a rare entity, and the occurrence of Bartter syndrome along with Takotsubo syndrome makes it a bizarre incidence. Diagnosis of both is very crucial and important, as the signs and symptoms tend to be different for each patient. Here, the authors present a case of Takotsubo cardiomyopathy with acquired Bartter-like phenotype and colonic stricture. It was promptly diagnosed and well managed at first incidence, but failing a timely follow-up led to mortality of patient, which highlights the importance of follow-up.
Key Points
1. Takotsubo syndrome is often encountered and is mistaken as acute coronary syndrome; however, management is different in both scenarios.2. This case report describes a rare association of Takotsubo cardiomyopathy with electrolyte imbalance, which clinically resembled Bartter syndrome. The patient’s condition improved following electroylte management and conservative treatment.
3. An early rule-out of acute coronary syndrome is important because management of the two
conditions differs but mortality is similar.
INTRODUCTION
Takotsubo syndrome (TTS), also known as stress-related cardiomyopathy or apical ballooning, is a transient systolic and diastolic dysfunction that is often preceded by some physical or emotional triggers. First described in Japan in 1990, it is derived from a Japanese word meaning ‘octopus pot’.1 The incidence of stress cardiomyopathy occurs in 1–2% of patients with ST-elevation myocardial infarction.2 Bartter syndrome is a rare autosomal recessive disorder affecting the function of the thick ascending limb of the loop of Henle, which causes salt wasting and hypokalaemic metabolic alkalosis.3 Adult-onset Bartter syndrome is rare and has been reported to be associated with chronic sialadenitis, pulmonary tuberculosis, and exposure to aminoglycosides.
CASE REPORT
This is an interesting case of a 56-year-old female diagnosed with a colonic stricture, which was likely inflammatory in nature. They presented with a history of decreased appetite, abdominal distension for the previous month, on-and-off loose motions, and vomiting for the last 10 days. The patient was admitted to the medical ward with the above symptoms, as well as generalised weakness and a confused state. Although there was no chest pain, an ECG was completed which showed acute ST elevation in the anterior leads. They were immediately referred to the cardiac centre in view of acute coronary syndrome.
On admission to the coronary care unit, the authors found marked ST elevation in V1–V6 leads (Figure 1). Cardiac biomarkers were positive, and the echocardiogram showed an akinetic apex in the anterolateral area. The patient’s blood pressure on presentation was 90/70 mmHg. The arterial blood gas test showed respiratory alkalosis with severe hypokalaemia and hypocalcaemia.
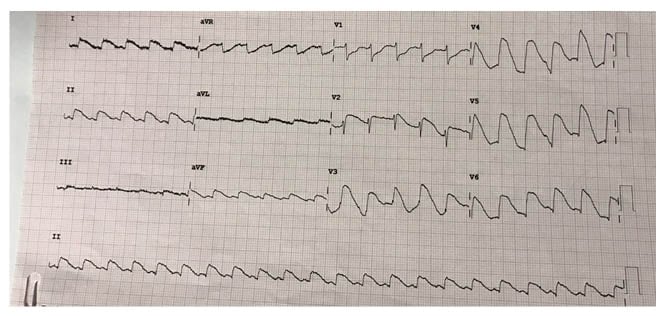
Figure 1: ECG during admission.
The authors planned for an immediate coronary angiography and revascularisation, and unexpectedly, thrombolysis in myocardial infarction Grade 3 flow was observed in all coronaries (Figures 2 and 3). Following this observation, it was thought that the patient was displaying with either myocarditis or Takotsubo cardiomyopathy. In the absence of fever, the authors made a provisional diagnosis of Takotsubo cardiomyopathy and stopped antiplatelets, heparin, and rapidly tapered inotrops. The patient’s blood pressure remained at 90/60 mmHg. The authors gave antidepressants, but persistent electrolyte disturbance occurred.
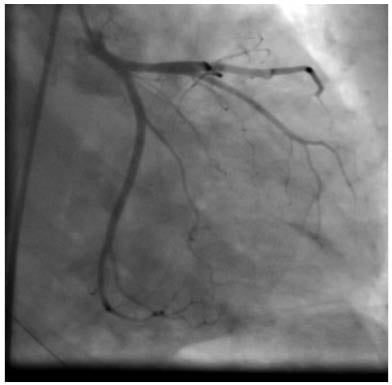
Figure 2: Normal angiogram of left anterior descending and left circumflex arteries during admission.
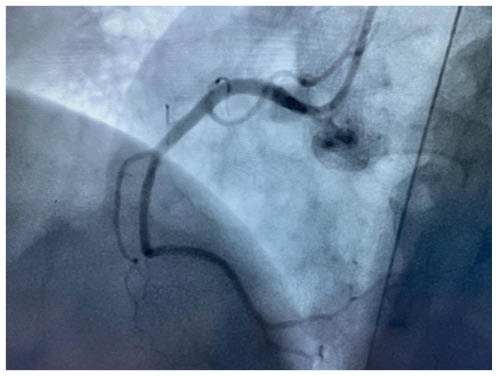
Figure 3: Normal angiogram of right coronary artery during admission.
Upon further evaluation, the patient presented with mild hypomagnesaemia, hyponatraemia, hypochloraemia, elevated renin, and elevated aldosterone, all suggesting secondary hypoaldosteronism. Following further analysis, the electrolyte abnormalities matched the Bartter phenotype. The authors’ administered a potassium supplement and spironolactone, with continued monitoring and treatment. Within the next 24 hours, ECG changes began showing improvement and both the ECG (Figure 4) and echocardiogram normalised within next couple of days.
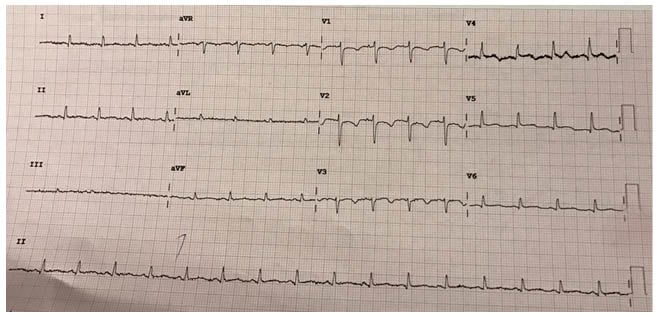
Figure 4: Normal ECG findings at Day 4.
The patient was discharged and advised to attend a regular follow-up. However, the patient did not attend the planned follow-up visit, and 1 month later they developed similar symptoms and succumbed with probability of a similar disease, precipitating due to poor compliance. No autopsy was performed.
DISCUSSION
The underlying mechanism of Takotsubo cardiomyopathy remains unknown. Some studies have pointed to the pathophysiological role of catecholamine, which may cause multivessel coronary spasm or diffuse coronary vasoconstriction.4 Post-menopausal females are most commonly affected, with clinical and ECG features indistinguishable from acute coronary syndrome. The most common complaint is often chest pain. Generally, some kind of physical or emotional trigger precedes presentation of TTS in patients.5 It is important to differentiate primary versus secondary diagnosis of TTS, as outcomes for a secondary diagnosis reflect underlying acute medical illness and carry worse short- and long-term prognoses.
In TTS, an increase in levels of troponin and creatine kinase is low compared with the observed amount of regional wall motion abnormality and left ventricular (LV) dysfunction. Increases in brain natriuretic peptide and N-terminal pro-brain-type natriuretic peptide are also higher than that of troponin and creatine kinase.6,7 Coronary angiography is characteristically normal, with an incidental finding of coronary disease in occasional patients. However, through LV angiography and transthoracic echocardiography, TTS can be distinguished as one of the following five types: apical, midventricular, basal, focal, and global.8 For treatment of this entity, judicious use of fluids, β-blockers, and angiotensin-converting enzyme inhibitors are advised, but the use of inotropes is generally contraindicated.9 An intra-aortic balloon pump may worsen LV outflow obstruction. There is high recurrence rate of 4%.10
Bartter syndrome results from the mutation of the five ion transport proteins in the thick ascending limb of the loop of Henle, resulting in hyponatraemia, hypokalaemia, hypochloraemia, metabolic alkalosis, hypocalcaemia, and mild hypomagnesaemia, as well as increased urinary prostaglandin excretion, plasma renin, and aldosterone levels.11 Recently, a similar phenotypic picture has also been reported in adults in cases of chronic sialadenitis,12 pulmonary tuberculosis,13 and in those with gentamicin exposure.14 It presents with polyuria, polydipsia, and salt craving. Treatment includes vitamin K and magnesium supplements as well as increased salt intake. The addition of spironolactone and amiloride can be appropriate.
CONCLUSION
In the authors’ case, the emotional stress superimposed upon the colonic stricture may have resulted Takotsubo cardiomyopathy and acquired Bartter-like syndrome. As no diuretic abuse or persistent vomiting was noted, it was considered to be an acquired idiopathic Bartter-like phenotype. The simultaneous presence of TTS and Bartter-like phenotype has never been observed, and both may have been precipitated because of stress. The possibility of an association between the inflammatory colonic stricture and stress cardiomyopathy could not established. The authors’ patient was treated with vitamin K supplements, spironolactone, and antidepressants. Dramatic improvement was noted in the clinical picture, ejection fraction, and electrolyte imbalance.