BACKGROUND AND AIMS
Eyelid dermatitis represents a unique diagnostic challenge due to the thin periocular skin, increased permeability, and frequent exposure to topical agents, which amplify inflammatory responses even at low levels of exposure.1,2 While cosmetics are well-recognized triggers, over-the-counter (OTC) ophthalmic drops remain an underrecognized cause of both allergic and irritant contact dermatitis, often resulting in delayed diagnosis and inappropriate management.1-3
This review characterizes the epidemiology, causative agents, pathophysiology, diagnostic challenges, and management implications of eyelid contact dermatitis associated with OTC ophthalmic drops.
MATERIALS AND METHODS
A focused literature review was conducted using PubMed and Embase, emphasizing preservative systems, active pharmaceutical ingredients, and diagnostic strategies including standard patch testing and patient product (‘as is’) testing.1
Allergic contact dermatitis from OTC ophthalmic drops is primarily a Type IV delayed hypersensitivity reaction mediated by T lymphocytes, typically presenting 24–72 hours after exposure.3 Preservatives, such as benzalkonium chloride, play a central role by disrupting epidermal barrier integrity while simultaneously promoting sensitization, leading to overlapping irritant and allergic mechanisms.4,5 Common offending agents include preservatives (benzalkonium chloride, thimerosal, parabens), antibiotics (neomycin, bacitracin), and excipients such as propylene glycol, many of which have well-documented sensitization potential (Table 1).1
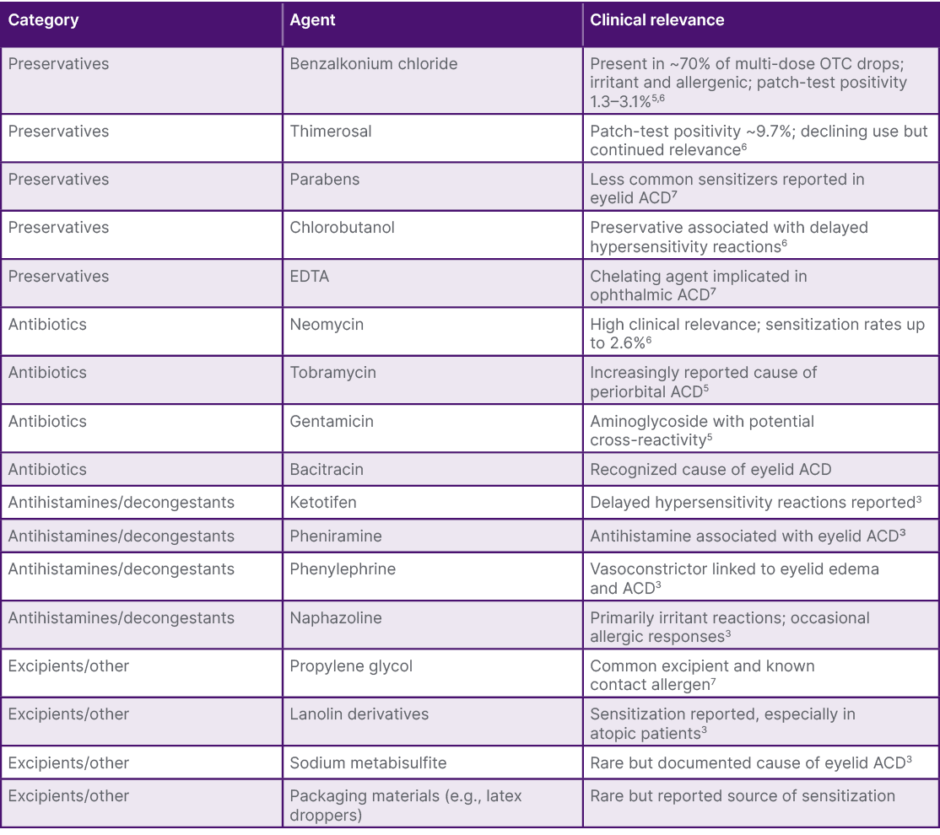
Table 1: Common offending agents in over-the-counter ophthalmic drops associated with eyelid contact dermatitis.3,5-7
ACD: allergic contact dermatitis; EDTA: ethylenediaminetetraacetic acid; OTC: over-the-counter.
BACKGROUND AND AIMS
Eyelid dermatitis represents a unique diagnostic challenge due to the thin periocular skin, increased permeability, and frequent exposure to topical agents, which amplify inflammatory responses even at low levels of exposure.1,2 While cosmetics are well-recognized triggers, over-the-counter (OTC) ophthalmic drops remain an underrecognized cause of both allergic and irritant contact dermatitis, often resulting in delayed diagnosis and inappropriate management.1-3
This review characterizes the epidemiology, causative agents, pathophysiology, diagnostic challenges, and management implications of eyelid contact dermatitis associated with OTC ophthalmic drops.
MATERIALS AND METHODS
A focused literature review was conducted using PubMed and Embase, emphasizing preservative systems, active pharmaceutical ingredients, and diagnostic strategies including standard patch testing and patient product (‘as is’) testing.1
Clinically, patients present with pruritic, erythematous, and edematous plaques involving the upper and/or lower eyelids, often with a characteristic ‘tear-drip’ distribution.6 Conjunctival involvement may result in chronic blepharoconjunctivitis. Importantly, patients frequently fail to recognize OTC ophthalmic drops as potential triggers unless specifically questioned, contributing to diagnostic delay.3 Diagnostic pitfalls commonly arise when clinicians focus exclusively on cosmetic exposures or rely solely on standard patch testing, which may fail to identify ophthalmic-specific allergens.1,3
RESULTS
Identification and avoidance of the offending agent remain the cornerstone of management.5,7 Preservative-free or single-use ophthalmic formulations are preferred for patients requiring chronic therapy. Acute flares may be treated with low-potency topical corticosteroids, used cautiously in the periocular region. Early dermatologic involvement facilitates targeted allergen identification, improves counseling, and enables interdisciplinary coordination with ophthalmology to prevent recurrent exposure.7
Despite increasing recognition, OTC ophthalmic drop-induced eyelid dermatitis remains a diagnostic blind spot. Barriers include low clinical suspicion, overlapping irritant and allergic mechanisms, and incomplete allergen identification with standard testing approaches. Expanded diagnostic strategies, including ophthalmic-specific patch testing and direct product testing, may improve diagnostic accuracy and patient outcomes.6,7
CONCLUSION
In conclusion, OTC ophthalmic drops are a frequent yet underrecognized cause of eyelid contact dermatitis. Preservatives and topical antibiotics represent the most common culprits, with benzalkonium chloride playing a key role in barrier disruption and sensitization. Early recognition, targeted history-taking, and appropriate testing are essential to reduce morbidity and avoid unnecessary therapeutic escalation.1