Abstract
AIDS is defined by several opportunistic infections, including Kaposi sarcoma (KS) and tuberculosis. This report describes a complicated case of AIDS-associated Kaposi sarcoma with syphilis and cutaneous tuberculosis coinfection. A 32-year-old bisexual male presented with a 3-month history of violaceous patches initially on the right sole spreading to the whole body. Cutaneous examination showed violaceous to brownish patches and plaques on the face, hard palate, trunk, both upper and lower extremities, with a nodule on the right sole. He was also diagnosed with gastrointestinal tuberculosis with a finding of a mesentery abscess positive for acid-fast bacilli stain. Sexually transmitted diseases laboratory work-up revealed late latent syphilis treated with benzathine penicillin G, and AIDS with a CD4 count of 29 cells/µL, and was started on anti-retroviral medications. Skin punch biopsy of the patch on the right thigh with CD31 and CD34 stain confirmed KS. While on antiretroviral and antituberculosis medications, the patient developed scrofuloderma. Skin punch biopsy of the right neck showed cutaneous tuberculosis with KS features. After 5 months of antiretroviral therapy and imiquimoid 5% cream as local therapy, there was 50% improvement in cutaneous lesions, and the patient was then lost to follow-up. A high index of suspicion and a thorough examination should be done in this population for early diagnosis and management of these complications.
Key Points
1. Advanced AIDS is a diagnostic challenge. Profound immunosuppression (CD4 count <30) allows for the simultaneous occurrence of multiple opportunistic pathologies. A high index of suspicion must be upheld by the clinician in order to personalize treatment strategies for each patient.2. This case is of a 32-year-old male presenting with disseminated Kaposi sarcoma, late latent syphilis, and gastrointestinal tuberculosis. It also highlights the clinical challenge of immune reconstitution inflammatory syndrome, where the patient’s improving immune system, following antiretroviral therapy, paradoxically triggers a new inflammatory response in the form of cutaneous tuberculosis (scrofuloderma).
3. Clinicians must perform thorough examinations, as Kaposi sarcoma can coexist with other infections. Patients should be monitored for immune reconstitution inflammatory syndrome and visceral involvement. A multidisciplinary approach involving dermatology, oncology, and infectious diseases is recommended. Treatment plans should also focus on wellness and prevention for a more holistic approach.
INTRODUCTION
HIV is part of the Retroviridae family of viruses, which induces suppression of the immune system and dysregulation primarily via depletion of CD4+ lymphocytes.1 AIDS is the late stage of HIV, which can be diagnosed when CD4 cell counts plummet to levels of <200 cells/µL or infections caused by opportunistic pathogens are identified, including both Kaposi sarcoma (KS) and Mycobacterium tuberculosis (M. tuberculosis) infection.1 One of the most frequently seen cancers among individuals affected by HIV is KS,2 occurring mostly in homosexual or bisexual men with abnormally low levels of CD4 count, usually below 350 cells/mm.1,3 It is an angioproliferative disorder arising from endothelial cell infection by KS Herpesvirus or Human Herpesvirus-8 (HHV-8).4 M. tuberculosis, on the other hand, causes an endemic pulmonary infection and is a major public health concern in the Philippines.5 Extrapulmonary tuberculosis, a less common form of M. tuberculosis infection, occurs in patients infected with HIV. Seventy percent of individuals afflicted by tuberculosis and concomitant AIDS infection are usually affected.6 Lastly, syphilis is a venereal disease primarily caused by Treponema pallidum, a part of the Spirochaetaceae family of bacteria.7 A resurgence of this bacterial infection has caused it to be identified as a global health risk, especially among individuals who are HIV-positive.8 Due to risk-taking behaviors such as sexual intercourse without the use of condoms among the younger men who have sex with men (MSM) population, concomitant infection of syphilis among the HIV afflicted patients is increasing.8
This study describes a case of a Filipino bisexual male, who was HIV-positive with co-existing KS, syphilis, gastrointestinal, and cutaneous tuberculosis.
CASE SUMMARY
This is a case of a 32-year-old, bisexual male from Manila, Philippines, who presented with a 3-month history of multiple violaceous patches and plaques starting on the right sole, gradually spreading to the face, upper and lower extremities, with no other associated symptoms such as pain or pruritus.
Two months prior to the consult, the patient also had fever, body malaise, right lower quadrant pain, anorexia, and vomiting. He was admitted under an impression of appendicitis, hence underwent exploratory laparotomy and appendectomy. Intraoperatively, a mesentery abscess was seen, which revealed 3+ for acid-fast bacilli stain. Hence, an assessment with gastrointestinal tuberculosis was made, and he was started on an anti-tuberculosis single-tablet regimen: rifampicin 150 mg, isoniazid 75 mg, pyrazinamide 400 mg, ethambutol 275 mg once daily. There was relief from the abdominal symptoms.
One month prior to the consult, the patient had intermittent fever and was admitted for sexually transmitted diseases workup at a tertiary government hospital. He tested positive at both screening and confirmatory testing for HIV with a CD4 cell count of 29 cells/µL. Further laboratory work-ups were done, revealing non-reactive Rapid Plasma Reagin (RPR) and hepatitis B surface antigen (HBsAg), while the treponemal test (TPPA) was positive. He was started on a fixed-dose combination of lamivudine 300 mg, efavirenz 600 mg, and tenofovir disoproxil fumarate 300 mg for HIV-1 treatment. Benzathine penicillin G 2,400 IU was administered weekly intramuscularly in the buttocks for 3 doses as treatment for late latent syphilis. Prophylaxis for opportunistic infections was also started, including fluconazole (cryptococcosis) and cotrimoxazole (Pneumocystis pneumonia). The patient was referred to the dermatology service for further evaluation of the skin lesions.
The patient works as a civil engineer. He is single, with several sexual partners, including 3 female and 12 male partners, and engages in unprotected oral, anal, and vaginal sexual intercourse wherein he assumes both penetrative and receptive roles. The review of systems was unremarkable. The patient’s medical and family history were non-contributory.
Cutaneous examination showed violaceous to brownish patches and plaques on the tip of the nose, preauricular and temporal area, left clavicular area, both arms and thighs, palms, soles, and hard palate (Figure 1). Dermoscopy shows a homogeneous pattern consisting of white lines on a pink to brown background on the patches and plaques, and a rainbow pattern on the nodule (Figure 2). Skin punch biopsy, CD34, and CD31 staining showed KS (Figure 2).
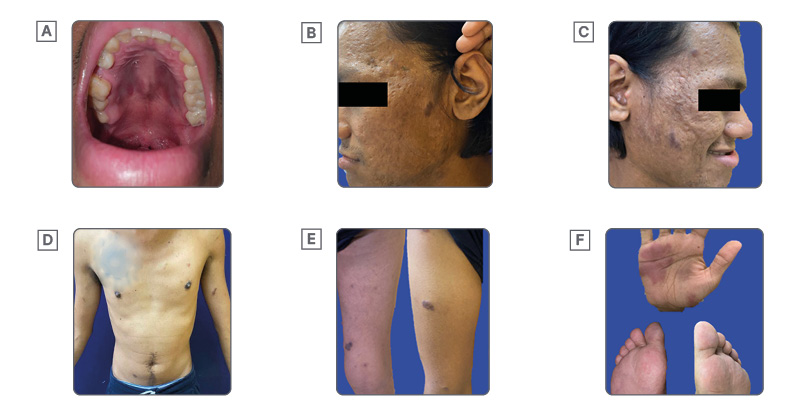
Figure 1: Clinical presentation of Kaposi sarcoma in a 32-year-old male.
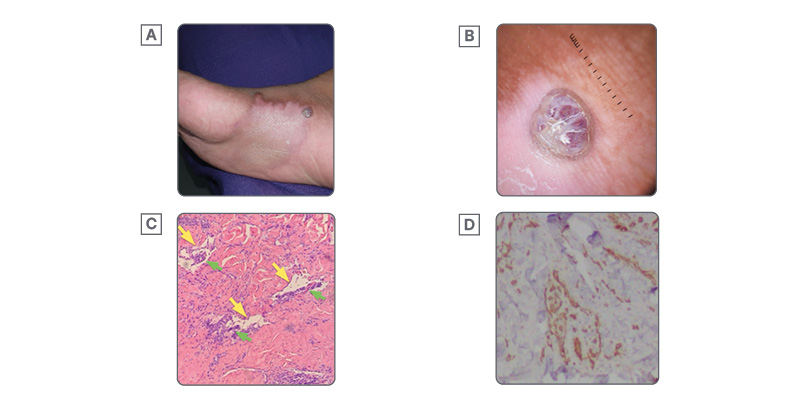
Figure 2: Dermoscopy, histopathology and, immunohistochemistry of Kaposi sarcoma.
The patient was lost to follow-up for 3 months but continued his antiretroviral and anti-tuberculosis medications. During the interim, the patient developed erythematous, non-painful nodules on both sides of the neck, which spontaneously ruptured. On physical examination, non-tender cervical lymphadenopathies were noted, and a cutaneous examination showed round to oval, erythematous, non-tender plaques with ulceration and purulent discharge with hemorrhagic crusting on the neck. The clinical impression was scrofuloderma; hence, Tissue Gene Xpert® (Cepheid, Sunnyvale, California, USA) of the neck was requested, which was negative. Wound Gram stain and culture and sensitivity were also negative. Another skin punch biopsy of the right neck was done, which had positive staining for acid-fast bacilli, CD34, and CD31, assessed as cutaneous tuberculosis with KS features (Figure 3).
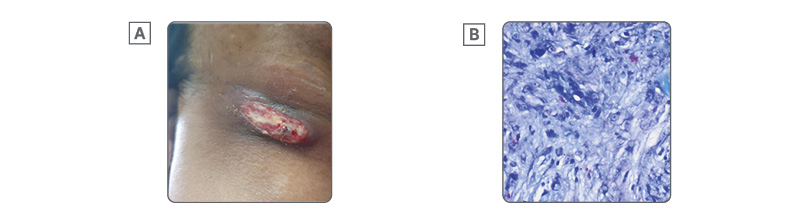
Figure 3: Scrofuloderma.
To assess the visceral involvement of KS, chest X-ray and fecal occult blood test were requested, which were both negative. A CT scan of the neck, chest, and whole abdomen was also requested; however, it was not done due to financial constraints. Imiquimod 5% cream applied three times weekly was started as local therapy for the cutaneous lesions.
After 5 months of antiretroviral therapy (ART), there was a 50% improvement in the lesions with noted flattening of the plaques and lightening of the violaceous color of some patches and plaques on the face, upper, and lower extremities. However, efforts have been made to reestablish communication, but the patient eventually discontinued follow-up. A detailed chronological summary of the patient’s clinical course, including presentation, diagnostic workup, and management, is presented in Table 1.
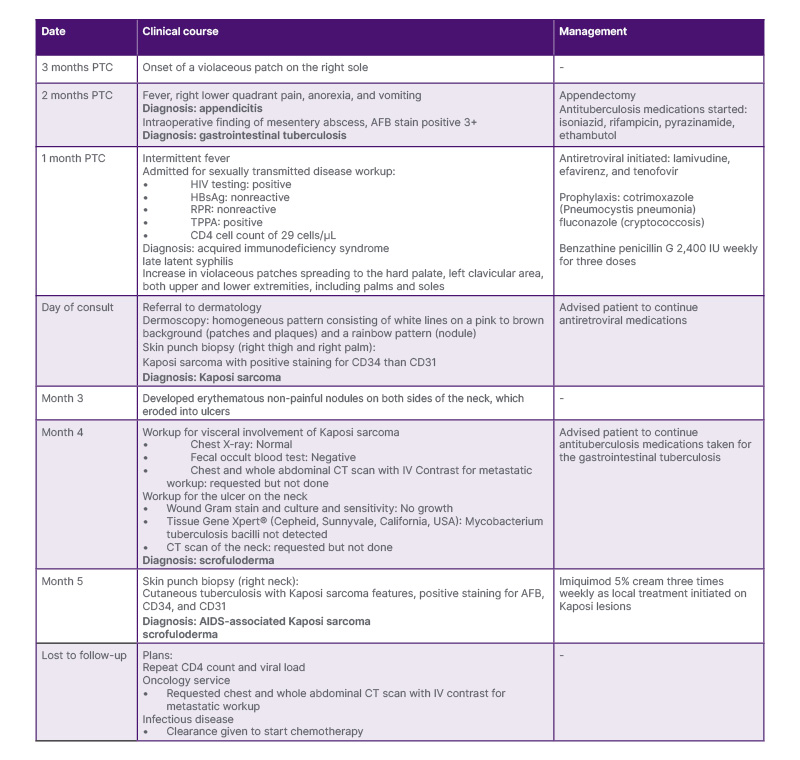
Table 1: Timeline of clinical course and management.
AFB: acid-fast bacilli; HBsAg: hepatitis B surface antigen; PTC: prior to consult; RPR: Rapid Plasma Reagin;
TPPA: Treponemal test.
DISCUSSION
Advanced HIV disease (CD4 count <200 cells/µL) remains a diagnostic challenge due to the coexistence of multiple opportunistic infections and malignancies.9 This case is particularly significant for its simultaneous presentation of KS, late latent syphilis, and extrapulmonary tuberculosis, making this case rare and of clinical value.
KS is a variety of an angioproliferative disorder stemming from infection of an endothelial cell by Kaposi sarcoma-associated herpesvirus (KSHV) or HHV-8.4 Hyper-proliferation of KSHV-infected cells is usually seen together with immunomodulatory cell infiltration, which results in chronic inflammatory states, angiogenesis, and tumor progression.4 This occurs statistically in 481.54 per 100,000 person-years among individuals who are HIV-positive, with the highest incidence observed in HIV-positive MSM.10 Local incidence based on the Philippine Dermatological Society Health Information System shows that there are 67 patients from 2011–2019 with KS, and the cases are predominantly in males.
There are 4 clinical and epidemiologic types:11,12 1) the classic type affecting older men of Mediterranean or Eastern European origin, which presents with a slow, indolent course with lesions commonly appearing on the lower extremities; 2) the endemic type occurs in sub-Saharan Africa and demonstrates a broader clinical spectrum, ranging from indolent cutaneous disease to aggressive forms involving lymph nodes and visceral organs, particularly among young adults and children; 3) the iatrogenic type arises in individuals receiving immunosuppressive therapy following organ transplantation and may show partial or complete regression upon reduction of immunosuppressive agents; and the last type 4) the epidemic type, which is associated with HIV/AIDS, present in the authors’ patient, represents the most aggressive form characterized by widespread mucocutaneous and visceral involvement due to profound immunosuppression.
The clinical manifestations of KS usually present with multiple, pigmented, non-blanching, painless macules and papules on the lower extremities.1 Larger oval plaques located on the torso often line up along the skin creases.13 Protruding, ulcerated, and hemorrhagic nodules can develop with edema associated with tenderness.13 AIDS-associated KS commonly presents with oral lesions in the palate, which is present in the authors’ patient, and may be one of the first symptoms of underlying immunosuppression.14 Visceral lesions are also more common among individuals with AIDS-related KS, frequently occurring in the lungs, lymph nodes, and gastrointestinal tract.15
A very important tool in dermatology in this era is dermoscopy. The rainbow pattern was seen in a nodular lesion using a contact polarized-light dermatoscope, a common finding in KS lesions.16 KS is commonly diagnosed based on the distinctive appearance of the lesions alone, but confirmation of the diagnosis must be based on histologic examinations.17 Histopathology usually shows spindle cells, growth of irregular vessels with slit-like configurations, extravasation of erythrocytes, and leukocytic infiltration encompassing full dermal thickness with associated plasma cells, and intra- and extracellular hyaline globules as well as the promontory sign (Figure 2).18
The histopathology result of the right neck of this patient showed cutaneous tuberculosis with KS features. In patients with HIV, more than one pathology in a single biopsy site can be seen, and case reports documented KS in association with tuberculosis, cryptococcosis, and Mycobacterium avium intracellulare in the same sample.19 A case report in Zimbabwe showed a 25-year-old patient who was HIV-positive whose biopsy of the mass of the right neck showed features of both cutaneous tuberculosis and KS, similar to this patient.20
Several markers can be used to differentiate KS from other vascular lesions.21 Latency-associated nuclear antigen (LNA1) is a highly sensitive and specific marker, given the strong correlation between HHV-8 and KS.21 However, since this is not available locally, the authors utilized other endothelial markers for vascular conditions, particularly CD31 and CD34, which both have positive staining in their sample. CD31 (platelet endothelial cell adhesion molecule-1) is considered one of the most sensitive and specific markers for endothelial differentiation; it highlights the spindle cells and abnormal vascular channels characteristic of KS, making it particularly useful in distinguishing it from other spindle cell neoplasms. CD34, another endothelial marker, is also commonly expressed in KS and stains both the tumor cells and surrounding vascular structures, although it is less specific because it can be positive in a wider range of soft tissue tumors.11 In practice, both CD31 and CD34 are used together as part of an immunohistochemical panel to support the diagnosis, typically alongside detection of HHV-8, which is more specific for KS.22 While CD31 offers higher specificity for vascular differentiation, CD34 provides complementary sensitivity, and their combined expression strengthens diagnostic confidence, especially in early or atypical lesions.11,22 In advanced stages of KS, CD34 exhibits stronger immunoreactivity than CD31, which was seen in the case.17
The AIDS Clinical Trial Group (ACTG) of the National Institute of Health developed the most frequently used staging system for AIDS-related KS.23 There are three categories in this staging, including tumor, immune system, and systemic disease. The patient in this case had lesions confined to the skin with minimal oral disease, CD4 count of <29 cells/µL, and systemic disease, showing a history of opportunistic infection, particularly extrapulmonary tuberculosis, but with the Kanofsky score of 90 defined as capable of normal activity but with few symptoms or signs of the disease T0I1S1. Pending the metastatic workup, the author’s patient falls into poor-risk.
Based on the NCCN Clinical Guidelines in Oncology, AIDS associated KS can be classified as having limited or advanced cutaneous disease.23 Asymptomatic individuals who are cosmetically unremarkable are considered to have limited cutaneous disease, and these patients may be treated with ART alone.23 As observed in the patient, there was 50% improvement on the patches and plaques with 5 months of ART.
Topical imiquimod has been investigated as a local therapy for KS, particularly for cutaneous and localized lesions.23-27 Imiquimod acts as a toll-like receptor 7 agonist, inducing local production of cytokines such as interferon-α and promoting antitumor immune responses as well as antiangiogenic effects.24 A systematic review of topical therapies for KS identified imiquimod as one of the most commonly studied agents, where 5% imiquimod cream was typically applied for several weeks (median duration approximately 17 weeks), sometimes under occlusion.24 Clinical outcomes showed complete response in approximately 34.6% of patients and partial response in 30.8%, with overall good tolerability and mainly mild local adverse effects such as erythema and pruritus.24 Individual case reports further support its efficacy. One study reported complete clearance of classic KS lesions after 20 weeks of imiquimod 5% cream applied three times weekly under occlusion, with no recurrence at 6-month follow-up.25 Another case demonstrated near-complete regression of lesions within 12 weeks, again with minimal toxicity.26 Additional reports have shown successful treatment of localized genital KS, highlighting its usefulness as a noninvasive option in anatomically sensitive areas.27 Available studies suggest that topical imiquimod is a promising, well-tolerated local therapy for selected patients with limited cutaneous KS; hence, this was also used for this case.
The prognosis of KS varies widely and depends on several factors, particularly immune status, extent of disease, and response to therapy.28 The introduction of ART has significantly improved survival in AIDS-associated KS by promoting immune reconstitution and reducing viral replication, which can lead to stabilization or regression of lesions. Patients with limited disease, higher CD4 counts, and absence of systemic symptoms generally have better outcomes.23 The determinants significantly associated with increased mortality or the necessity for chemotherapeutic regimens include tumor Stage of T1, a decreased CD4 count of less than 200 cells/µL, and a positive test for HHV-8.28 As with the case, a CD4 count of 29 cells/µL puts the authors’ patient at poor risk.
Scrofuloderma is an unusual variety of extrapulmonary tuberculosis, encompassing 1–2% of cases, but it is the most common form of extrapulmonary tuberculosis in patients who are HIV-positive and in the pediatric age group.29 While on treatment, this patient also developed scrofuloderma signifying immune reconstitution inflammatory syndrome (IRIS), which occurs in 18% of patients who are HIV-TB co-infected.30 Occult tuberculosis was subsequently unmasked in this patient as the immune system recovered, presenting with multiple neck nodules. IRIS is a paradoxical exacerbation of a pre-existing infection or disease process or the emergence of a new infection/disease process soon after initial therapeutic modalities.1 Upon initiation of ART, individuals with concurrent infections of HIV-1 and tuberculosis (TB) are vulnerable to developing TB-associated IRIS (TB-IRIS).31 Patients with low CD4+T cell counts at the time of ART initiation, followed by a rapid increase in CD4 counts post-ART, are more likely to develop TB-IRIS.31
On the sexually transmitted disease workup for this patient, a diagnosis of late latent syphilis was made through serology. The presence of syphilis alongside TB and KS further complicates this case. Recent data shows a rising trend in syphilis and HIV co-infection, with approximately 45.5% of high-risk populations showing positive serology for both.32 Syphilis is a venereal disease with protean manifestations stemming from Treponema pallidum infection. Systemic illness is of initial consideration due to vasculitis as its primary pathology.33 With negative non-treponemal test RPR and positive treponemal test TPPA, this patient was diagnosed with the late latent syphilis34,35 and underwent treatment with benzathine penicillin G 2,400 IU weekly for three doses based on the CDC Sexually Transmitted Guidelines 2015.36 Monitoring should be done using RPR, and there should be a four-fold decrease in titers to determine if the patient had been treated completely.36 Hicks et al.37 described a repeated non-reactive Veneral Disease Research Laboratory Test (VDRL) and Fluorescent Treponemal Antibody Absorption (FTA-ABS) in an HIV-positive individual with concomitant Kaposi’s sarcoma and secondary syphilis. Impairments in antibody responses among HIV afflicted individuals are shown by various studies as well. As seen in these cases, the antigenic response may range from complete seronegativity to exaggeration inconsistent with the clinical findings.38
HIV is associated with many opportunistic infections and malignancies, and the presence of syphilis and TB coinfections with KS makes this a rare case. A similar case described a 30-year-old Indian male with HIV, with generalized purplish or brown rash, where histopathology of cutaneous lesions confirmed the diagnosis of KS.39 During workup, he was also positive for syphilis, pulmonary tuberculosis, and primary effusion lymphoma. A high index of suspicion for early diagnosis and long-term monitoring must be advised to these patients.
Ethics and Consent
Written informed consent was obtained from the patient for publication of this case report and accompanying clinical images. Patient anonymity was strictly maintained, and no identifying information was disclosed in this manuscript.
CONCLUSION
This is a complicated case of AIDS associated KS with extrapulmonary tuberculosis and syphilis. This case differs from previous literature by highlighting the prognosis and clinical improvement despite its quadruple presentation. Timely intervention and a multidisciplinary approach are necessary. Clinicians must always maintain a high index of suspicion in patients who are severely immunocompromised.