Meeting Summary
A satellite symposium on the evaluation and management of dysphagia in children was held during the 12th Annual Congress of the European Society for Swallowing Disorders (ESSD) in Leuven, Belgium, on 14th September 2022. Dysphagia is the impairment or difficulty in swallowing and may have an oropharyngeal or oesophageal cause. This can result in delayed transit of liquids or solid food from the mouth to the stomach. Food thickening agents can reduce regurgitation and improve swallowing mechanics, particularly in infants and young children. At this symposium, Karen van Hulst, Speech and Language Therapist and Clinical Epidemiologist at Radboud University Medical Center, Amalia’s Children’s Hospital, the Netherlands, introduced the topic of dysphagia and its complications, and the evaluation of dysphagia. She then discussed the use of thickening agents in children with dysphagia. Ben Hanson from the Department of Engineering, University College London, UK, introduced the International Dysphagia Diet Standardization Initiative (IDDSI). The IDDSI has developed a standardised and practical method of measuring the thickness of liquid and food that can be used when preparing foods at home or in the hospital, and may be applied commercially. Analou Sugar, Clinical Lead in Paediatric Speech and Language Therapy at Chelsea and Westminster Hospital Foundation Trust, London, UK, ended the symposium with a presentation of her recent clinical experience using a new thickener (ThickenUp® Junior, Nestlé Health Science, Vevey, Switzerland) in paediatric patients.
Introduction
Dysphagia is defined as “any disruption to the swallow sequence that results in compromise to the safety, efficiency, or adequacy of nutritional intake.”1 Dysphagia may have an oropharyngeal or oesophageal cause, resulting in delayed transit of liquids or solid food from the mouth to the stomach.1 Approximately one in 100 children experience swallowing difficulties; therefore, the clinical evaluation and management of abnormalities in swallowing are important for adequate nutrition and the health of children.2 Three symposium speakers at the 12th Annual ESSD Congress, Karen van Hulst, Ben Hanson, and Analou Sugar, introduced the topic of paediatric dysphagia, its assessment, and management using dietary thickeners.
Introduction to Paediatric Dysphagia and the Importance of Thickeners
Karen van Hulst
Problems Arising from Dysphagia
To enable safe and efficient feeding, a child should be able to swallow effectively. “Appropriate nutritional input is essential for development and growth during infancy and childhood,” said van Hulst, and “feeding should be pleasant, tasty, social, joyful, and provide opportunities for social interaction.”
The complex swallowing process starts with two voluntary phases: oral bolus preparation and oral bolus propulsion. These phases are followed by the involuntary pharyngeal phase, where food is irreversibly directed into the oesophagus and the airway is automatically protected from aspiration; and the oesophageal phase, where food is moved down to the stomach.1
Paediatric dysphagia can occur due to a disorder in one or more of these phases, and can manifest as an inability to swallow food, drinks, or medication; or regurgitation of reflux material or saliva, also called posterior drooling. Dysphagia can result in direct or indirect aspiration before, during, or after swallowing. Aspiration prior to swallowing is most common when drinking liquids as the bolus may go from the mouth to the pharynx before airway closure occurs. Aspiration during swallowing is most often due to co-ordination problems. Aspiration after swallowing is usually caused by a pharyngeal bolus residue, most likely due to a problem with swallow strength.1
Dysphagia can arise at different levels, all with their own swallow patterns, and can be due to central neurological problems, such as spasticity or central hypotonia syndromes; bulbar motor problems; muscle disorders; or anatomical problems.1 While an estimate of approximately 1.0% of the general paediatric population may have swallowing difficulties,2 incidence is much higher in at-risk populations.3 For example, 90.0% of children with neurological impairments have oropharyngeal dysfunction, 80.0% of children with developmental delays and 24.5% of premature infants have feeding problems, and 22.0−50.0% of children with cardiopulmonary disease have dysphagia.4
In neuromuscular disorders, muscle weakness can result in pharyngeal residue following swallowing. Children with central nervous system disorders such as cerebral palsy can develop dysphagia in one or more phases of swallowing.5 The findings from a study of 130 preschool children with cerebral palsy showed that common observed signs during mealtimes included coughing (44.7%), multiple swallows (25.2%), a gurgly voice (20.3%), wet breathing (18.7%), and gagging (11.4%).6
Dysphagia can not only prolong mealtimes, lead to an inability to consume foods and fluids safely and in adequate quantities, and cause respiratory problems; it can also have longer term consequences such as impaired growth due to inadequate nutrition, and lowered health outcomes and quality of life, with increased stress for the child and their caregivers.7,8
Assessment of Dysphagia
The ‘Algorithm for Evaluation and Management of Paediatric Dysphagia’ provides a stepwise approach for clinical assessment. Evaluation first involves distinguishing between feeding difficulties due to dysphagia or oral aversion, specific food aversion, or other symptoms. If dysphagia is suspected, the next stage is to evaluate whether there is an oropharyngeal or oesophageal cause.1 This is most often carried out using a video fluoroscopic swallow test, which should be personalised through discussion with the child’s caregiver(s), clinical team, and the radiology department.1,4 Diagnosis in children with complex feeding problems may also involve endoscopic evaluation of swallowing. Here, a flexible scope is passed through the nose so that the area above the vocal folds can be viewed and swallowing visualised.1
Management of Dysphagia
Treatment for paediatric dysphagia depends on the underlying cause. For example, a child with oral dysphagia may require feeding therapy, e.g., changing feeding position, pacing feeds, and/or conditioning exercises; and/or changing a feeding bottle nipple to one that best suits the condition. A child with pharyngeal dysphagia associated with aspiration of food and drink may require surgical management (gastrostomy) as well as medical management, including using thickening agents (thickeners) combined with feeding therapy and/or formula changes. Treatment for oesophageal dysphasia may require either medical or surgical management, depending on the cause of the condition.4,9,10
Children with central and peripheral neurologic disorders have distinctive patterns of dysphagia when they undergo a videofluoroscopic swallowing examination. For instance, children with cerebral palsy tend to have more problems with thin liquids, whereas children with spinal muscular atrophy tend to have more problems with puréed foods.5 This means, advised van Hulst, that while thickeners should be used for children with cerebral palsy if co-ordination is a problem, for children with neuromuscular disorders, thickeners should preferably not be used if swallow strength is a problem.
Food and thickeners are commonly used for children with dysphagia. They have been shown to enable better oral co-ordination, improve pacing, change swallow mechanics, and allow the bolus to move more slowly from the oropharynx to the oesophagus. The use of thickeners for feeds has also been shown to reduce the number of regurgitation episodes in several studies, and is supported by gastroesophageal reflux disease (GERD) guidelines from the North American Society For Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN), and the European Society for Paediatric Gastroenterology Hepatology and Nutrition (ESPGHAN).11,12
There is a range of different thickeners available for the paediatric population, and van Hulst recommended using a thickener that does not change the nutritional value or composition of the food it is added to. Food purées, for example, often contain fructose, and infant cereals can be high in iron and carbohydrates. Starch-based thickeners can change viscosity over time when they come in contact with saliva, as well as being broken down by amylase. Locust bean and xanthan gum-based thickeners have an advantage by not having any of these issues.11
Van Hulst noted that while thickened liquids reduce the risks of laryngeal penetration and aspiration, they can increase the risk of post-swallow residue in the pharynx. Thickened food and liquids also require greater strength in terms of tongue propulsive forces used to drive a bolus to the oropharynx. If these propulsive mechanisms are reduced, and there is also reduced pharyngeal strength, there is a risk for residues to remain in the pharynx after swallowing, increasing the risk of post-swallow aspiration.9,11 Therefore, she advised, thickeners should not be used in children with reduced pharyngeal muscle strength.
Van Hulst concluded by discussing that thickeners are usually well-tolerated with few side effects. However, close follow-up is required to ensure that children tolerate thickeners, and that there is an adequate improvement in the symptoms of dysphagia.
Optimal Food and Beverage Thickening: Introduction to the International Dysphagia Diet Standardization Initiative (IDDSI) Standards
Ben Hanson
Assessing the Texture of Foods and Drinks
The IDDSI framework is a continuum that can be used to rate consistency levels of food or drink.13 Figure 1 shows that drinks can be rated from 0 to 4 (thin to extremely thick) and foods from 3 to 7 (moderately thick to regular/easy to chew).13
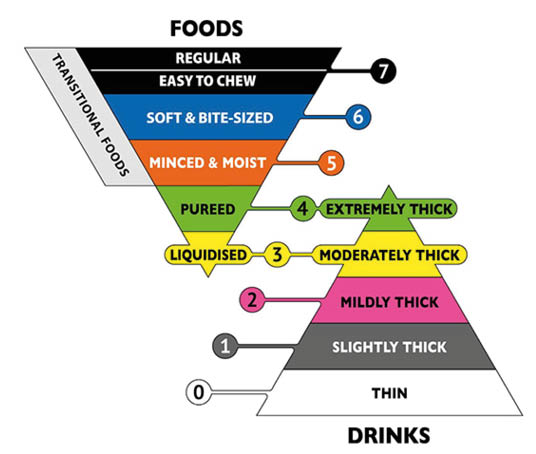
Figure 1: The International Dysphagia Diet Standardization Initiative (IDDSI) framework. Food and drinks classification and testing.
The International Dysphagia Diet Standardisation Initiative 2019 at https://iddsi.org/framework/ Licensed under the CreativeCommons Attribution Sharealike 4.0 License https://creativecommons.org/licenses/by-sa/4.0/legalcode. Derivative works extending beyond language translation are NOT PERMITTED.15
These IDDSI ratings consider the food breakdown process, from taking a bite to producing a smooth, lump-free, moist bolus capable of being swallowed, as well as physiological information regarding tracheal size, particle size, and bolus smoothness.13
In the paediatric population, increasing food texture is fundamental to the normal developmental process of weaning a child onto solid foods. Texture, said Hanson, can include particles, crunchiness, slipperiness, and thickness. The latter is a quality that can be used to modify the swallowing process. Echoing the study presented by van Hulst, Hanson commented on a study related to intra-oral pressures with IDDSI-standardised textures in healthy adults. This study showed that increasing the thickness of a liquid enhanced the pressure used by the tongue to swallow boluses. This pressure increase reduced the volume of sip size participants took, which, said Hanson, illustrates how changes in thickness can have a variety of influences on the swallowing process.14
Hanson also discussed how, as infants expend a lot of energy in the process of eating, drinking, and digesting, there is a need to make sure this process is not impeded in children with dysphagia, as increasing the fluid thickness increases the amount of energy needed to extract liquids through a nozzle due to the above discussed pressure increase.14 However, while increasing feeding bottle teat hole size may be seen as a solution, this could actually increase the risk of aspiration.11
Ensuring the Optimal Use of Thickeners
There is a long history in food science of controlling texture using a range of ingredients. Thickeners are predominantly composed of powdered carbohydrate grains. When added to a liquid, these swell up and partially dissolve, forming a gel network that holds the liquid together, thus increasing thickness.16 Hanson mentioned, in the question and answer session, that thickener can be added to hot or cold drinks, and that for powdered drinks it is best to mix them up first, then add a thickener.
The thickening process can be influenced by a variety of factors. For instance, when trying to thicken milk, the presence of fats, proteins, and sugars can inhibit the absorption of water and with some thickeners it can take at least 30 minutes, and up to 24 hours, for a stable thickness to be reached.17 Additionally, the method used to mix up liquids, be it stirring or shaking, can also have a large effect on the end result when using a thickener.18 In the question and answer session, Hanson said: “The best way we found to produce a stable consistency quickly was to shake it in a bottle.” He explained that attention also needs to be paid to small changes in the thickness of a liquid that can arise when a measuring scoop is not levelled off properly, or the volume of the liquid to be thickened is not accurately measured each time.
Viscosity can be difficult to measure. Although some liquids may be Newtonian, such as honey or oils, most foods and drinks are non-Newtonian fluids. Yoghurt, for example, has properties of both a liquid and a solid. This makes it difficult to classify thick liquids consumed by children with dysphagia. In Hanson’s laboratory, a rheometer is used to measure samples under controlled conditions, but this is not a practical tool for a domestic or hospital/care home kitchen.16 Therefore, a flow test for liquids, based on IDDSI standards, has been developed, whereby thickness can be measured by assessing flow rate through a 10 mL syringe or a standardised IDDSI funnel.
For foods and thicker liquids at IDDSI Level 3 and above, the qualities of a thickened liquid can be assessed by ascertaining whether or not it drips off of a fork or slides off a spoon as a cohesive lump.13 To ascertain the softness of a food, Hanson’s team developed tests based on the pressure used to squash a food sample with a thumb on a fork where the nail blanched. This was found in laboratories to be roughly equivalent to the pressure the tongue uses to generate the swallow reflex. According to Hanson, these practical, easily reproducible tests can help ensure that a person with dysphagia is consuming foods and liquids that are easy to control on the tongue without spilling, and are moist and easy to swallow.
As an example, Hanson discussed the use of ThickenUp Junior, a thickener composed of 10:1 locust bean gum and xanthan gum.19 Unpublished results he presented showed that when added to a commercial ready-to-use baby milk formula (SMA Pro Follow-On Milk 2®, Nestlé Health Science, Vevey, Switzerland) this can reach a stable consistency very quickly when thoroughly mixed, and remain stable over 24 hours, which is important when feeds are interrupted (Figure 2).
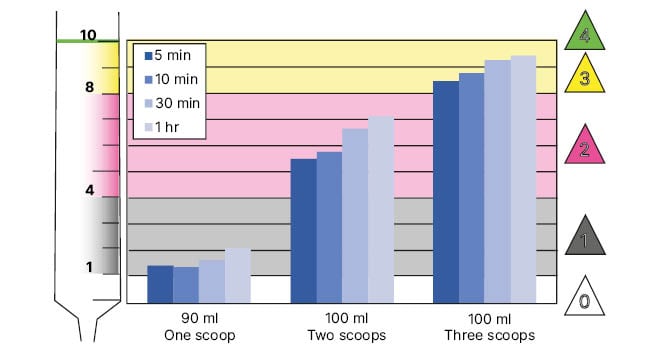
Figure 2: Thickness over time when adding a defined amount of ThickenUp® Junior to defined volumes of a commercial infant formula (SMA® [SMA Nutrition, Nestlé, Vevey, Switzerland] Pro Follow-On Milk 2; scoops).
Hanson also reported that similar results had been found using a wide range of milks and soft drinks, such as orange juice or chamomile tea, and even high energy powdered formula (for example, SMA Pro Follow-On Milk 2). He showed that small changes to formula/water mixing ratios, for instance using 90 mL rather than 100 mL of water, can allow for fine-tuning of a liquid’s consistencies to meet specific needs (Figure 3).
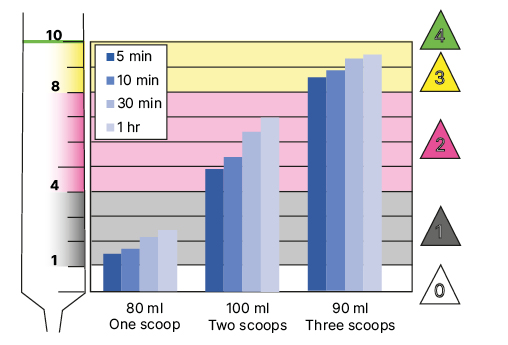
Figure 3: Thickness over time of defined ratios of a commercial infant formula (SMA Pro Follow-On Milk 2®, Nestlé Health Science, Vevey, Switzerland) and water when mixed with a defined amount of ThickenUp® Junior (scoops).
Hanson also added that he had noted how adding a small amount of saliva can change the consistency of a food such as porridge made with milk, decreasing its IDDSI level. He found that adding an amylase resistant thickener (such as ThickenUp Junior) could prevent changes due to saliva.
Using the IDDSI tests, peer-reviewed, scientific studies are now building a consensus as to how dysphagia can be managed with texture modification. These include investigations of transitional food samples, and studies of how modification techniques impact the rheological properties of foods and liquids used for people with dysphagia.20,21 Together these mean, that in domestic and hospital or care home settings, a standard thickness of a particular food or liquid can be produced every time it is needed.
In conclusion, Hanson discussed the need to consider how food and drink texture affects an individual’s swallowing, as this can impact the comfort of the child, their quality of life, and their safety.11 The IDDSI standards can be used to help select products and diets, monitor consumption progress, and aid research in dysphagia.
Tolerance and Effectiveness of a New Paediatric Thickener: Clinical Experience
Analou Sugar
Sugar began her presentation by stating: “The impact of dysphagia on a parent and family should never be underestimated.” She continued: “It’s very disempowering if you can’t feed your child or if you have to be socially excluded, be different, struggle with feeds, etc.” In support of these statements is a recently published cross-sectional study that included 50 children aged 2–5 years with a diagnosis of feeding disorder. The findings showed that paediatric feeding impairments can impact areas such as social, physical, emotional, and school functioning of the child; and daily activities, feeding, and worry for the caregiver. Importantly, the impact of poorly managed dysphagia on the wellbeing of children and their families was more significant than for several other paediatric conditions, including kidney transplantation and acute liver failure. The authors concluded that improved diagnosis and treatment individualised to each child’s needs could improve the overall wellbeing of children and their families.7
To illustrate the utility of thickeners, Sugar reported the results of a trial of ThickenUp Junior that aimed to evaluate its acceptability, gastrointestinal (GI) tolerance and compliance, in children aged 6 months and above for the management of reflux, GERD, and dysphagia. The trial ran from August 2021 to May 2022 and recruited UK National Health Service (NHS) patients in both inpatient and outpatient settings with a diagnosis of dysphagia and under the care of a multidisciplinary team. Parents completed a 28-day diary reporting the amount of thickener used, amount of liquid consumed, and tolerance to the feeds.
GI tolerance was determined by recording several GI markers. These included symptoms such as diarrhoea, constipation, bloating/distension, nausea, vomiting, burping, flatulence, and regurgitation; and those that may be associated with GI symptoms, such as indices of abdominal discomfort and pain, back arching, and crying. The Bristol Stool Chart22 was used to describe stool consistency.
Inclusion criteria were infants and children who were already using a thickener or required such for management of GERD and/or dysphagia, and who were able to comply with the study protocol. Exclusion criteria were contraindications to any of the ingredients used in ThickenUp Junior or milk, as some milk products may be present during processing of the thickener. During the trial, no new weaning or introduction of new foods could take place. Parents were trained on how to use ThickenUp Junior to prepare liquids and provided with written information regarding preparation of liquids at home.19
Of the 15 children included in the study, eight were female, seven were male, and the age range was 9 months–8 years, with 10 participants under the age of 32 months. Participants presented with a range of diagnoses, including asthma, bulbar palsy, congenital heart disease, prematurity, trisomy 21, GERD, and tracheoesophageal fistula. Six participants withdrew from the study, three due to refusing the new thickener, and three to becoming unwell for reasons unrelated to thickener use.
Drinks were prepared to a range of IDDSI levels from 1 to 3, with IDDSI 3 used more in the older children and IDDSI 1 and 2 in younger participants.
The results showed that all hydration requirements and targets were met or improved. The use of this new thickener was generally well-tolerated, with any reported GI symptoms being limited and transient. Three recruits became unwell and discontinued the study; this was not deemed to be study-related.
Sugar also discussed the study participants’ experiences, as reported by their caregivers. One said: “We have noticed that she is not vomiting or coughing when drinking, which is fab!” Another reported that the thickener “has made nursery staff and them [the caregivers] more confident, and they are ‘less scared’ due to no coughing or vomiting.” A third caregiver described how their child was “going to the drink station at nursery to grab her sippy cup, the one with a straw, and drink by herself.”
The limitations of the study were acknowledged during the question and answer session. These included the small number of study participants, and the lack of a control group. Sugar commented that this is a first promising trial and more assessment is needed. She suggested that further studies could utilise standardised measures of feeding and caregiver responses, and these could be monitored both pre-intervention and post-intervention to give a better idea of the effectiveness and experience using ThickenUp Junior.
In conclusion, Sugar discussed how using ThickenUp Junior meant that drinks can be prepared to a consistent IDDSI level, which is a safer practice, can aid compliance, and enhance the patient experience. She reported that her findings show that the use of ThickenUp Junior can enhance the patient’s experience, and she suggested that, even-though not firmly investigated yet, its use may reduce the need for hospital appointments and interventions, such as enteral feeding and management of GI complications in children with dysphagia.








