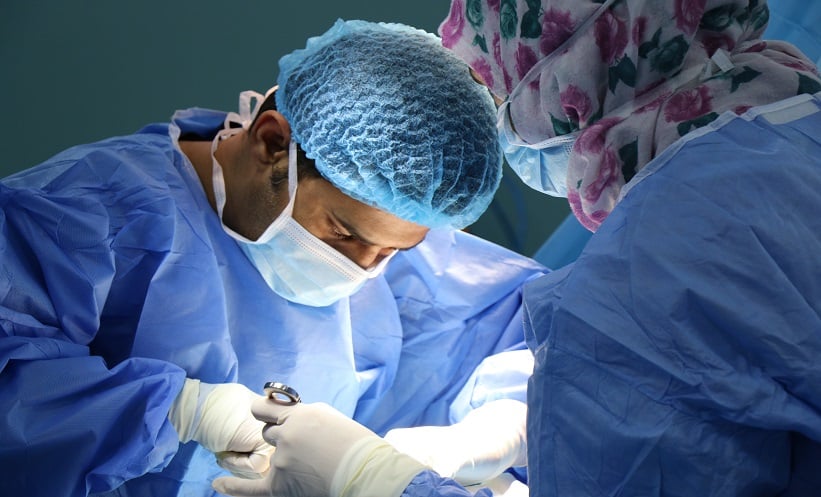
Sepsis poses a substantial public health challenge, affecting approximately 3.4 million individuals and causing 700,000 deaths every year in the World Health Organization (WHO) European Region.1 Early identification of the aetiological agent and appropriate antimicrobial therapy are crucial for improving the long-term outcomes of patients. A number of methods for rapid organism detection have been described in the literature. A study by Cordovana and Ambretti2 evaluated the rapid Sepsityper® kit (Bruker Daltonik GmbH, Bremen, Germany), using a large collection of routine positive blood cultures collected from February 2018 to October 2019, and found that this procedure demonstrated an excellent efficacy in detecting the most clinically relevant causative agents of sepsis. Furthermore, in the context of neonatal sepsis, haematological parameters and biomarkers may help to facilitate early recognition and treatment, thereby improving prognosis.
Clinical guidelines are essential for optimising the quality of care received by patients with sepsis. Since the publication of the 2016 Surviving Sepsis Campaign guidelines, additional evidence has been accrued, necessitating a revision of the recommendations in 2021. Changes to the current practices of screening and initial resuscitation were particularly evident in the updated document.3
In a Society of Critical Care Medicine (SCCM) media release,4 Waleed Alhazzani, Department of Medicine, McMaster University, Hamilton, Ontario, Canada, and Methodology Chair of the 2021 guidelines, highlighted one important aspect of the new recommendations: “A systematic applied screening process is essential to recognising patients with sepsis early.” Alhazzani added: “If you don’t look for it, you might miss sepsis, but the clock is ticking and time matters, so patients should constantly be reevaluated for where they are and what they need next.”
A strong recommendation, supported by moderate quality evidence, was issued against using the quick Sequential Organ Failure Assessment (qSOFA) score as a single screening tool. This was based on the low sensitivity of qSOFA, which uses a Glasgow Coma Scale (GCS) score under 15, a respiratory rate of at least 22 breaths/min, and a systolic blood pressure of 100 mmHg or less to identify patients with sepsis outside the intensive care unit setting. Research has determined that qSOFA is less sensitive than the Systemic Inflammatory Response Syndrome (SIRS) criteria when predicting risk. Moreover, only 24% of infected patients in the original derivation study had a qSOFA score of 2 or 3; however, these individuals were shown to account for 70% of poor outcomes. When comparing qSOFA against the Modified Early Warning Score (MEWS) and National Early Warning Score (NEWS), similar results have also been reported. Despite the poor performance of qSOFA, a positive score should still alert physicians to the possibility of sepsis.
The use of blood lactate measurements to screen for sepsis in adults with clinically suspected sepsis was made a weak recommendation, with a low quality of evidence, in the 2021 guidelines. A pooled analysis of three studies investigating the prognostic accuracy of lactate levels in sepsis found that this marker had a positive likelihood ratio of 4.75 and a negative likelihood ratio of 0.29. Studies have also shown that implementing point-of-care lactate testing at presentation is associated with reduced mortality. However, there are several drawbacks to this approach, ultimately limiting its usefulness as a clinical tool. For example, in resource-constrained environments, serum lactate testing might not always be available. Furthermore, measuring lactate on its own is neither sensitive nor specific enough to rule the diagnosis of sepsis either in or out.
For individuals with sepsis-induced hypoperfusion or septic shock, the 2016 guidelines strongly recommended administering at least 30 mL/kg of intravenous crystalloid fluid within the first 3 hours of resuscitation. This was downgraded to a weak recommendation in the 2021 guidelines. There was low quality evidence for both the 2016 and 2021 recommendations. A retrospective cohort study of adults with sepsis or septic shock revealed that failure to receive 30 mL/kg of crystalloid therapy within the first 3 hours of diagnosis was associated with increased odds of prolonged intensive care unit stay as well as in-hospital mortality. Pre-randomisation, the average fluid volume received in the ProCESS, ARISE, and ProMISe trials was in the range of 30 mL/kg, indicating that this volume has now been adopted in routine practice. However, to date, there have been no prospective interventional studies that compare different volumes for initial resuscitation.
Ensuring that guidelines for the management of sepsis and septic shock are regularly updated to reflect the most up-to-date evidence and clinical information will enable healthcare professions to provide the best care. Accurate and current guidelines will also empower patients to make informed healthcare choices, which take into account their personal preferences and needs. This will be crucial to improving outcomes and ending preventable deaths from sepsis.
References
1. World Health Organization (WHO). Hand Hygiene Day: it’s in your hands – prevent sepsis in health care. 2018. Available at: https://www.euro.who.int/en/health-topics/Health-systems/health-workforce/news/news/2018/5/hand-hygiene-day-its-in-your-hands-prevent-sepsis-in-health-care. Last accessed: 14 March 2022.
2. Cordovana M, Ambretti S. Direct matrix-assisted laser desorption ionisation (MALDI) identification from positive blood cultures: rapid Sepsityper®. EMJ Microbiol Infect Dis. 2020;1(1):29-31.
3. Evans L et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock 2021. Crit Care Med. 2021;49(11):e1063-143.
4. Society of Critical Care Medicine (SCCM). Updated adult sepsis guidelines emphasize treatment of long-term consequences of illness. 2021. Available at: https://www.sccm.org/getattachment/About-SCCM/Media-Relations/Surviving-Sepsis-Campaign-Adult-Guideline-2021.pdf?lang=en-US. Last accessed: 24 March 2022.