INTRODUCTION
Heart failure (HF) is a leading cause of morbidity.1 It results from impairment in ventricular filling or relaxation and can be broadly classified as heart failure with preserved ejection fraction (HFpEF) or with reduced ejection fraction (HFrEF).2 Strategies for preventing HF are paramount. Prevalent coronary artery calcium and extracoronary calcification are associated with future coronary heart disease3-7 and HF8 events. Less is known about the impact of progression of valvular calcification (VC) (mitral annular [MAC] and aortic valve calcification [AVC]) on HF risk.
OBJECTIVE
The authors assessed the prospective association between VC progression and incident HF and its subtypes, as well as indices of left ventricular (LV) structure and function.
METHODS
The Multi-Ethnic Study of Atherosclerosis (MESA) is an ethnically diverse cohort of females and males who were free from clinical cardiovascular disease and HF at baseline. VC progression (interval change of >0 units/year) was assessed by two cardiac CT scans over a median of 2.4 years and was defined as 0 for no progression, 1 for progression at only one valve site (AVC or MAC), and 2 for progression at both sites (AVC and MAC). The authors also assessed progression at each individual valve site creating a binary variable, i.e., AVC/MAC progression versus no AVC/MAC progression. Cox regression was used to determine the risk of adjudicated HF and linear mixed effects models to determine 10-year change in LV parameters measured by cardiac MRI associated with VC progression. HFpEF was defined as having an ejection fraction ≥50% and HFrEF was defined as <50%.
RESULTS
The study included 5,591 MESA participants with a mean (± standard deviation) age of 62 (±10) years, 53% were women, and 40% of participants were Caucasian. Twelve percent had prevalent AVC and 9% had prevalent MAC, while 10% had AVC progression and 10% had MAC progression. Eighty-three percent had no VC progression, 15% progressed at only one site (AVC or MAC), and 3% progressed at both sites. There were 251 incidents of HF over 15 years. After adjusting for cardiovascular risk factors, the hazard ratios (HR) and 95% confidence interval (CI) of HF associated with VC progression at one and both sites were 1.62 and 1.21–2.17, and 1.88 and 1.14–3.09, respectively, compared with no progression. HR were stronger for HFpEF (HR: 2.52; 95% CI: 1.63–3.90 and HR: 2.49; 95% CI: 1.19–5.25) but not significant for HFrEF. Both AVC and MAC progression were associated with HF (HR: 1.61; 95% CI: 1.19–2.19 and HR: 1.50; 95% CI: 1.09–2.07, respectively), and with HFpEF (HR: 1.84; 95% CI: 1.16–2.94 and HR: 2.30; 95% CI: 1.47–3.59), respectively). There was worsening of some LV parameters over 10 years (Table 1).
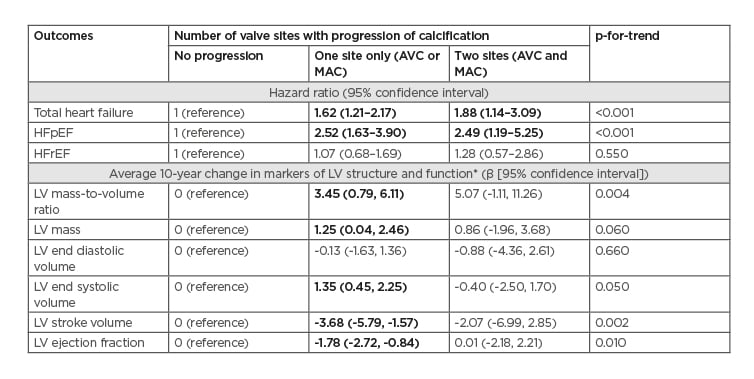
Table 1: Association between progression of valvular calcification, heart failure, and markers of left ventricular structure and function.
AVC: aortic valve calcification; HFpEF: heart failure with preserved ejection fraction; HFrEF: heart failure with reduced ejection fraction; LV: left ventricular; MAC: mitral annular calcification.
*Analysis limited to 2,748 participants with cardiac MRI data.
Bolded results are statistically significant, p<0.05.
Results are adjusted for age, race/ethnicity, sex, MESA site, CT scanner type, time between baseline and follow-up CT scans, educational status, BMI, smoking status, pack-years of smoking, physical activity, systolic blood pressure, use of antihypertensives, use of lipid lowering medications, total cholesterol, HDL-cholesterol, diabetes status, and estimated glomerular filtration rate.
CONCLUSION
The authors found VC progression over a median of 2.4 years to be associated with higher risk of HF, HFpEF, and various indices of LV structure and function. There was no significant association with HFrEF and LV end diastolic volume. It is unknown if interventions aimed at reducing VC progression will reduce HF and HFpEF risk.








