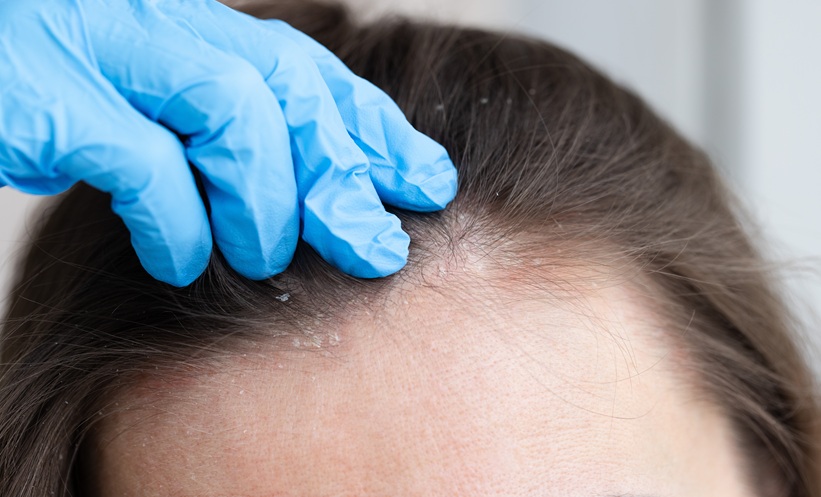
A 52-WEEK analysis of a Phase 3b trial has found that obesity did not significantly affect the efficacy or safety of tildrakizumab in patients with moderate-to-severe scalp psoriasis. The findings add to growing evidence that biologic therapies targeting interleukin (IL)-23 can remain effective in patients with obesity, despite concerns that excess adipose tissue and chronic inflammation may reduce treatment response.
Researchers evaluated outcomes in patients receiving tildrakizumab 100 mg versus placebo, stratified by obesity status, defined as a body mass index (BMI) of at least 30 kg/m². Approximately half of participants in both treatment groups were classified as having obesity at baseline.
Obesity Did Not Impact Treatment Efficacy for Scalp Psoriasis
At Week 16, patients treated with tildrakizumab achieved significantly higher response rates than those receiving placebo, regardless of obesity status. Among patients with obesity, 48.8% achieved an Investigator Global Assessment modified 2011 (scalp) response, compared with 10.0% in the placebo arm. Similar results were observed in patients without obesity, with response rates of 50.0% versus 4.8%, respectively.
Comparable improvements were also seen in Psoriasis Scalp Severity Index (PSSI) 90 responses. In patients with obesity, 62.8% of those treated with tildrakizumab achieved at least 90% improvement in PSSI scores, compared with 10.0% receiving placebo. In patients without obesity, 58.7% achieved PSSI 90 responses, while no patients in the placebo group met this endpoint.
Additional benefits included reductions in scalp itch severity and decreases in affected scalp surface area. Investigators reported that treatment responses remained consistent through Week 52 in patients receiving tildrakizumab.
The safety profile was similar between patients with and without obesity, and no new safety signals were identified during the study period.
Findings Support Tildrakizumab Use in Difficult-to-Treat Scalp Psoriasis
Obesity is recognised as a major comorbidity in psoriasis and has previously been associated with lower treatment responses to some systemic therapies. The authors noted that obesity contributes to systemic inflammation, which may alter drug pharmacokinetics and therapeutic outcomes. However, the current findings suggest that tildrakizumab maintains durable efficacy in scalp psoriasis irrespective of BMI category.
The authors cautioned that the subgroup analysis was not prospectively powered to detect statistical differences between obesity groups. Nevertheless, they concluded that tildrakizumab demonstrated sustained efficacy and a consistent safety profile in patients with and without obesity over 52 weeks.
Reference
Sofen HL et al. Efficacy and safety of tildrakizumab for treatment of moderate-to-severe scalp psoriasis in patients with obesity over 52 weeks. Dermatol Ther (Heidelb). 2026;DOI: 10.1007/s13555-026-01774-2.
Featured image: Vitalina on Adobe Stock