INTRODUCTION
Diabetes is one of the most common chronic conditions. It affects a global population of approximately 425 million. The prevalence of diabetes is projected to increase to 629 million by 2045. The disease affects about 8.3% of adults worldwide, with the highest prevalence between the ages of 40 and 59 years.1 Diabetes is a progressive disease, which may lead to macrovascular, microvascular, and autonomic complications in the event of suboptimal control.2 There is convincing evidence that early and tight glycaemic control leads to better long- and short-term outcomes, including a reduction in long-term macrovascular and microvascular complications.3
Despite good-quality evidence and recommendations, glycaemic control is often shown to be inadequate on a global scale. Russell-Jones et al.4 showed that one-third of patients with Type 2 diabetes (T2D) in the UK failed to achieve target glycated haemoglobin test (HbA1c) levels of ≤7.5% (59 mmol/mol).
One of the most important causes for this is therapeutic inertia, previously known as clinical inertia, which is defined as the failure of healthcare professionals to advance or intensify treatment for patients with chronic conditions, despite available clinical guidelines.4,5 It not only happens in diabetes cases, but also in other chronic conditions such as hypertension and hyperlipidaemia.
CAUSES OF DIABETIC (THERAPEUTIC) INERTIA
Factors causative of therapeutic inertia can be divided into healthcare provider-related factors, patient factors, and organisational and systemic factors.
Healthcare provider-related factors contribute to 50% of the causes of therapeutic inertia.6 This includes time constraints, competing demands, inadequate training, lack of familiarity with the efficacy and safety of therapeutic regimens, lack of knowledge, and variations in guidelines and recommendations.7
Insulin is the most common and important therapy that healthcare professionals fail to initiate, intensify, and titrate.8 The main reason attributed to the inertia is the fear of its side effects, specifically hypoglycaemia and weight gain, in addition to concern regarding patient acceptability of insulin as an injectable agent. Approximately 30% of therapeutic inertia may be attributed to patient factors. This includes concern regarding potential side effects, non-adherence, misunderstanding of treatment regimens, limited doctor-patient communication, low level of education, and presence of acute and terminal illnesses.9
The third group of causes of therapeutic inertia is related to organisational and systemic factors and contributes to 20% of causes of therapeutic inertia. This includes limitation in supply of medications, costs of medications, lack of individualised guidelines for patients, and insurance coverage.
THE IMPORTANCE OF DIABETIC INERTIA
Diabetes is a chronic, progressive condition. Therapeutic inertia impacts the health of patients and leads to higher levels of HbA1c and increased macro- and microvascular complications. It is vital to overcome therapeutic inertia, as there are benefits in terms of improved management of diabetes and reduced long-term costs, which will improve patients’ outcomes and quality of life.10 There are potential national healthcare benefits, including improving patient group outcomes, welfare, and social costs. For this reason, the authors conducted this study as the first of its kind about this problem.
METHODS
This was a retrospective cohort study, which included patients with diabetes hospitalised in the Elhwari General Hospital, Benghazi, Libya. These patients received medical therapy for different reasons (chest infection, cerebrovascular accident, urinary tract infection, etc.) in a medical ward over a period of 9 months. The information was collected from medical records and discharge notes. Treatment regimen was assessed before and during hospitalisation and at discharge to detect any change.
The primary outcome was a change in pre-admission and outpatients’ prescriptions at hospital discharge, and the relation of this change to the type of diabetes, the type of diabetic treatment given before hospital admission, blood glucose at admission, pre-hospital or hospital HbA1c, and hospital hypoglycaemia.
The authors also examined the number of patients who had their treatment changed (intensified or decreased), and the reason behind the change, including admission blood sugar; pre-admission or in-hospital HbA1c; occurrence of in hospital hypoglycaemia, or admission with hypoglycaemia; and no clear causes for the change.
STATISTICAL ANALYSIS
A descriptive statistical analysis was carried out. Categorical variables were described with absolute and relative frequencies, and quantitative data were described as mean and standard deviation (SD), interquartile range, and median. The proportion of the study population with Type 1 diabetes (T1D) was presented as a per cent. Pearson chi-square tests were used to compare the proportions of patients with and without treatment intensifications. Independent student’s t-tests were used to compare the mean, while the Mann–Whitney U test was used to compare the median between patients with and without treatment intensification.
RESULTS
A total of 315 patients with diabetes were included in this study. There were 140 males (44%) and 175 females (56%). Patients with T1D represented 9%, while 91% of patients had T2D. The mean age was 60.25 years (male: 62.08 years; female: 58.78 years), and the age range was between 18 and 93 years. Hospital stay duration was 2–30 days. Mean disease duration was 11 years. HbA1c was available for 41 patients (13%) during their hospital admission, or within 90 days before hospitalisation. HbA1c ranged between 4.9–15.5% (mean HbA1c: 8.9) with significant difference (p=0.004) between T1D (SD: 11.02+/-2.94) and T2D (SD: 8.4+/-2.04), but not between gender (male SD: 9.0890+/-2.5856; female SD: 8.895+/-2.4449; p=0.8). Of all the patients, 35% were smokers, and 60% had other comorbidities such as hypertension, dyslipidaemia, and/or chronic kidney disease (Table 1).
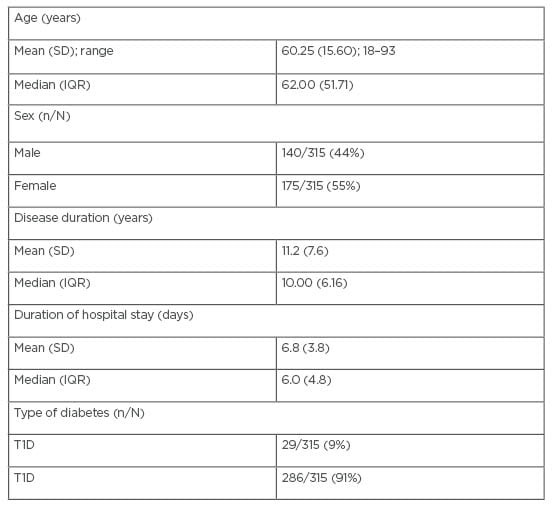
Table 1: Patients’ demographics.
IQR: interquartile range; SD: standard deviation; T1D: Type 1 diabetes; T2D: Type 2 diabetes.
According to treatment before admission, 132 patients (42.0%) were on oral hypoglycaemic and 134 (42.5%) were on insulin. Around one-fifth of the study population was on both insulin and oral hypoglycaemic medications. In addition, six patients were on diet control, and there were 22 patients with no treatment, most of whom had refused treatment before (Table 2).
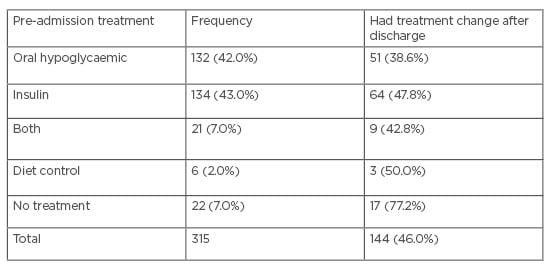
Table 2: Type of treatment at admission and changes after discharge.
Out of 315 patients with diabetes, 171 (54.28%) had no changes in their therapy during their hospital stay or even at discharge, while 144 patients had changes in their treatment plan. Of note, the predominant change was intensification of therapy (116 out of 144 patients). There were no changes in the management plan for 28 patients. The main factor of change was admission blood sugar, which occurred in 71 out of 144 patients (49.3%); 69 patients had treatment intensification as they came in with hyperglycaemia; and for two patients, the dose decreased as their admission blood sugar was low. On the other hand, 16 out of 28 patients’ treatment reduced at discharge as their admitted blood sugar was low. The second factor affected the change was the pre-admission or hospital HbA1c in 20 out of 144 patients (13.8%). Admission for hypoglycaemia constituted 11.8% (17 out of 144) of all causes of change. The majority of patients admitted with hypoglycaemia had a reduction of dose therapy, except one patient; surprisingly, the medical team intensified their treatment at the time of discharge without documenting the reason. The authors noticed that in a quarter of the patients (36 out of 144), there was no clear cause of change found in their files. The change in treatment according to the type of diabetes: 58.6% of patients had T1D and 44.4% had T2D (Table 3).
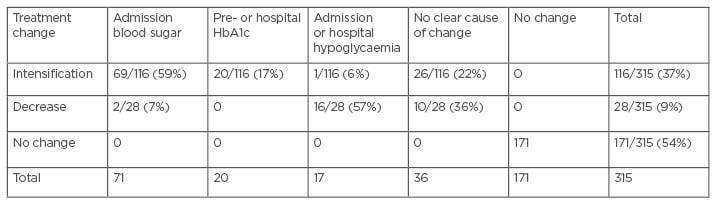
Table 3: Causes of treatment change.
HbA1c: glycated haemoglobin test.
Regarding factors that affected treatment change, the authors found a significant relation with gender (X2=6.159a;degrees of freedom: 2; p=0.046), and the type of treatment before admission (X2=27.176a; degrees of freedom: 8; p=0.001). These two factors affected the decision of the treatment change. There was no relation between treatment change with other factors such as age, HbA1c, type of diabetes, or the duration of hospital stay. Half of the patients who were admitted on diet control had been prescribed treatment after their discharge, while of the patients previously on no therapy, 17 had treatment after discharge, and five were discharged without treatment without clear cause (Table 4).
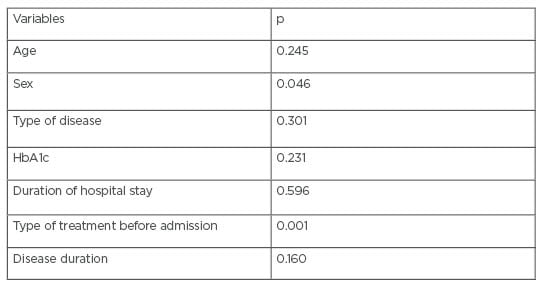
Table 4: Factors that affected treatment change.
HbA1c: glycated haemoglobin test.
DISCUSSION
The aim of this study was to assess the prevalence of diabetic inertia in one of the main hospitals in Libya and, to the best of the authors’ knowledge, this is the first study in this area.
The EDICT study demonstrated that the early intensive therapy of patients with newly diagnosed diabetes was more effective than conventional therapy.11 This study showed that using triple therapy with metformin, pioglitazone, and exenatide was superior to conventional stepped therapy with metformin, followed by the addition of sulfonylurea and insulin glargine. In the patients who received triple therapy, there was a significant reduction in HbA1c (5.95%), compared with the group who received conventional therapy (6.50%; p<0.001). Furthermore, even with the greater fall in HbA1c in the triple-therapy group, the incidence of hypoglycaemia was 7.5-fold lower than the conventional therapy group, despite having a greater reduction in HbA1c. One of the major consequences of therapeutic inertia is the extra cost on healthcare systems and public health due to the deterioration of patients who suffer from complications.12
The authors’ study confirmed the presence of therapeutic inertia in Elhwari General Hospital. The frequency of therapeutic inertia in this study was comparable with that found in previous studies. From their data, the authors found that there were around 36% of patients with uncontrolled diabetes who had treatment intensification and the main trigger for change was the admission blood sugar level. In comparison to other studies, such as El-Kebbi et al.,13 who showed that diabetes treatment was only intensified in 50% of patients with suboptimal T2D control. Similarly, the DICE study demonstrated that nearly 50% of patients with T2D in a primary care practice had an HbA1c above the target.14 Griffith et al.15 studied, suboptimally, patients with controlled diabetes with at least one hospital admission; they found that less than 25% of patients had treatment escalation or treatment change on their discharge, and few were allocated a follow-up visit. Therefore, there is compelling evidence that therapeutic inertia can lead to worse clinical outcomes.
In the authors’ cohort, the inpatient measure of HbA1c to determine glycaemic control was used in the minority of cases (13%). From hospital records, the authors found during the study period that the HbA1c test was available in the hospital. Physicians were aware about the availability of the test. When hospital physicians were asked why the measure of HbA1c was not undertaken in all in patients with diabetes, reasons offered included: the result may delay patient discharge and that diabetes was irrelevant to the reason for admission. A reason offered by the group as a reason for not intensifying treatment, despite a suboptimal HbA1c measurement, was waiting for confirmation of persistent suboptimal glycaemic control (a second consecutive HbA1c measurement above the target), before intensifying treatment when patients were close to their glycaemic target. The approach of postponing treatment intensification until after two consecutive measurements of above-target HbA1c may justify some cases of therapeutic inertia. However, a study by Sidorenkov et al.16 found that delay in therapy intensification due to waiting for two consecutive measurements of above-target HbA1c resulted in longer than recommended periods for treatment intensification for significant proportions of patients. Although a delay in therapy intensification may be justified for some patients, it took longer than recommended by existing clinical guidelines for significant proportions of patients to receive treatment intensification. Despite the availability of the test and results, other study groups faced the same issues of ignoring HbA1c as a useful resource.
Studies by López-Simarro,17 Vinagre et al.,18 and Gonzalez-Clemente et al.19 demonstrated that the lack of treatment intensification in patients with poor glycaemic control (HbA1c: ≥ 7%) varies between 32.2% and 52.5%.18,19 With regard to hospitalised patients, the comorbid prevalence of diabetes is around 35%, and the occurrence of hyperglycaemia during admission for non-diabetic reasons is associated with higher prevalence of morbidity and mortality.20 A large UK cohort study found that one year delay in treatment intensification significantly increased the risk of myocardial infarction, stroke, and heart failure by 67%, 51%, and 64%, respectively.21 Therefore, it is very important to address the issue of therapeutic inertia, and early and appropriate diagnosis and treatment of hyperglycaemia will lead to better health outcomes.
In contrast to other studies, the authors found no correlation between patient age, duration of hospital stay, or other comorbidities and the frequency of diabetic treatment intensification. Therapeutic inertia was present in this study in more than 54% of sampled patients. They also found treatment changes occurred, but without clear documentation for reason for change. In the majority of cases, no follow-up appointment for review of diabetic control was provided. Therefore, the authors think the cause of therapeutic inertia in their study was because of the healthcare provider, with some organisational factors.
Similarly with other studies, this study found that patients on more than one oral hypoglycaemic medication were less likely to be offered treatment intensification, despite an above-target HbA1c. Wan et al.22 similarly showed that incremental number of oral anti-diabetic medications were associated with reducing chance for treatment intensification. In comparison to patients with diabetes not on any pharmacological treatment, patients on dual or triple anti-diabetic therapy were 10 times less likely to receive treatment intensification.21 Importantly, the use of multiple medications can lead to low adherence, higher rates of side effects, and may incur additional cost to patients and health systems.
Adopting proactive approaches to prevent inertia such as routine clinical audits, patient feedback systems, consistent follow-up procedures, effective use of clinical information systems, education of healthcare professionals, and ease of availability of guidelines are ways to tackle this issue. The results of this study were discussed with the diabetes team in the hospital; they have taken some actions, such as better referral systems, and providing junior doctors with some educational resources.
RECOMMENDATIONS
The authors recommend that all patients with diabetes who are hospitalised for any reason should be referred to the endocrinology team or diabetes experts for review if suboptimal glycaemic control is suspected or confirmed, in order to improve hospital outcomes and long-term health status. Developing clear guidelines even at the level of the hospital for referral to diabetic services, systems to support identification and management of hyperglycaemia, and the education of medical and nursing staff, by providing frequent lectures and workshops in order to raise the awareness about therapeutic inertia and their causes, is important. Having clear post-discharge follow-up systems and routine patient education about diabetes and its complications, in addition to giving adequate time for each patient, would also be helpful. This will reduce the resistance for future escalation of treatment and allow the monitoring of therapies and their side effects. HbA1c has been proved to be useful in disease monitoring and gives precious information in terms of disease progression and prognosis, so it should be used when available.
LIMITATIONS FOR THE STUDY
The lack of an organised electronic system, as most of the data was extracted from paper notes, meant that some records could not be accessed or were missing, and the authors struggled to collect the complete data. Moreover, some discharge notes were poor quality (i.e., illegible handwriting or the document was damaged) and, therefore, extraction of records was incomplete in some cases. As in other retrospective studies, some statistics could not be measured, or were confusing.
CONCLUSION
The authors’ study shows that diabetic treatment inertia at a major tertiary hospital in Benghazi was high amongst patients with diabetes admitted to medical wards (54.28%). The therapeutic inertia in the study was likely due to doctors rather than the system or patients. HbA1c was carried out for only 13% of patients at admission and 25% (36 out of 144) had changes; however, there was no clear cause of change in their files. Patients on more than one oral hypoglycaemic medication were less likely to be offered treatment intensification, despite an above-target HbA1c. This work needs to continue with more patients in multicentre longitudinal studies.